Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 53(5); 2019 > Article
-
Original Article
Clinicopathological Characterization and Prognostic Implication of SMAD4 Expression in Colorectal Carcinoma -
Seung-Yeon Yoo1,2
 , Ji-Ae Lee1,2
, Ji-Ae Lee1,2 , Yunjoo Shin2
, Yunjoo Shin2 , Nam-Yun Cho2
, Nam-Yun Cho2 , Jeong Mo Bae,1,2
, Jeong Mo Bae,1,2 , Gyeong Hoon Kang,1,2
, Gyeong Hoon Kang,1,2
-
Journal of Pathology and Translational Medicine 2019;53(5):289-297.
DOI: https://doi.org/10.4132/jptm.2019.06.07
Published online: June 24, 2019
1Department of Pathology, Seoul National University Hospital, Seoul, Korea
2Laboratory of Epigenetics, Cancer Research Institute, Seoul National University College of Medicine, Seoul, Korea
-
Corresponding Author: Jeong Mo Bae, MD, Department of Pathology, Seoul National University College of Medicine, 103 Daehak-ro, Jongno-gu, Seoul 03080, Korea Tel: +82-2-2072-3374, Fax: +82-2-743-5530, E-mail: 'jeongmobae@gmail.com'
Corresponding Author: Gyeong Hoon Kang, MD, Department of Pathology, Seoul National University College of Medicine, 103 Daehak-ro, Jongno-gu, Seoul 03080, Korea Tel: +82-2-740-8263, Fax: +82-2-765-5600, E-mail: 'ghkang@snu.ac.kr'
© 2019 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- SMAD family member 4 (SMAD4) has gained attention as a promising prognostic factor of colorectal cancer (CRC) as well as a key molecule to understand the tumorigenesis and progression of CRC.
-
Methods
- We retrospectively analyzed 1,281 CRC cases immunohistochemically for their expression status of SMAD4, and correlated this status with clinicopathologic and molecular features of CRCs.
-
Results
- A loss of nuclear SMAD4 was significantly associated with frequent lymphovascular and perineural invasion, tumor budding, fewer tumor-infiltrating lymphocytes, higher pT and pN category, and frequent distant metastasis. In contrast, tumors overexpressing SMAD4 showed a significant association with sporadic microsatellite instability. After adjustment for TNM stage, tumor differentiation, adjuvant chemotherapy, and lymphovascular invasion, the loss of SMAD4 was found to be an independent prognostic factor for worse 5-year progression-free survival (hazard ratio [HR], 1.27; 95% confidence interval [CI], 1.01 to 1.60; p=.042) and 7-year cancer-specific survival (HR, 1.45; 95% CI, 1.06 to 1.99; p=.022).
-
Conclusions
- We confirmed the value of determining the loss of SMAD4 immunohistochemically as an independent prognostic factor for CRC in general. In addition, we identified some histologic and molecular features that might be clues to elucidate the role of SMAD4 in colorectal tumorigenesis and progression.
- Patients and tissue samples
- Under the exclusion criteria described previously [34], 1,370 out of 1,853 CRC cases resected at Seoul National University Hospital, Seoul, Korea, between January 2004 and June 2008 were reviewed. Among them, 1,281 cases with formalin-fixed, paraffin-embedded (FFPE) tumor blocks sufficient for construction of a tissue microarray (TMA) were included in the study. Initial pathologic diagnosis and clinical information, including age, tumor location and radiologic/pathologic evidences of distant metastases were obtained from electronic medical records. Additional histologic parameters including tumor differentiation, tumor budding, representative number of tumor-infiltrating lymphocytes (TILs) per one high power field (400× magnification), were evaluated by two pathologists as described previously.34 FFPE tissues were used for molecular analysis and immunohistochemistry.
- Immunohistochemistry
- For each CRC case, a pair of 2-mm cores of representative tumor areas in the FFPE tissue were extracted to construct the TMA. To evaluate SMAD4 expression in CRCs, TMAs were sectioned at a thickness of 4-μm and stained using a rabbit monoclonal anti-SMAD4 antibody (1:200 dilution, clone EP618Y, Abcam, Cambridge, UK). Stained slides were scanned by an Aperio AT2 slide scanner (Sausalito, CA, USA) at 40× magnification with a resolution of 0.25 μm per pixel. The proportion and the intensity of SMAD4 staining in nuclear compartment were evaluated using TMA Assistant protocol of QuPath [35], open-source software for digital pathology image analysis. In detail, the intensity of SMAD4 staining in nuclear compartment was graded using intensity feature of nuclear diaminobenzidine (DAB) optical densities (OD). Because nuclear DAB ODs of entrapped nonneoplastic epithelium range from 0.2 to 0.6, intensity of nuclear SMAD4 staining of tumor cells were evaluated using following cut-offs: intensity 0, DAB OD<0.2; intensity 1, DAB OD≥0.2 and <0.6; and intensity 2, DAB OD≥0.6. Finally, each case was classified to SMAD4-loss (≥95% of tumor cells showed intensity 0), SMAD4-low (≥5% of tumor cells showed intensity 1, and <30% of tumor cells showed intensity 2), and SMAD4-high (≥30% of tumor cells showed intensity 2). For survival analysis, CRC cases were dichotomized to CRCs with SMAD4 loss and CRCs with retained SMAD4 (SMAD4-low and SMAD4-high). The evaluation method and cut-offs for the expression of cytokeratin 7 (CK7) (clone OV-TL, 12/30, Dako, Carpenteria, CA, USA), cytokeratin 20 (CK20) (clone Ks20.8, Dako), and nuclear protein CDX2 (clone EPR2764Y, ready-to-use, Cell Marque, Rocklin, CA, USA) were described previously [36]. All immunohistochemical procedures in this study were conducted with an automated immunostainer (BenchMark XT, Ventana Medical Systems, Tucson, AZ, USA).
- Molecular analyses
- Through histological examination, representative tumor portions were marked and then subjected to manual microdissection. The dissected tissues were collected into microtubes containing lysis buffer and proteinase K and were incubated at 55°C for 2 days. DNA from paraffin-embedded tissues was extracted, and the polymerase chain reaction (PCR) was performed. Direct sequencing of KRAS codons 12 and 13 and allele-specific PCR for BRAF codon 600 were performed as described previously [34]. The MSI status of each tumor was determined by evaluating five microsatellite markers (BAT25, BAT26, D2S123, D5S346, and D17S250) as standardized by the National Cancer Institute [37]. A fluorescent label was added to either the forward or reverse primer for each marker, and the PCR products were electrophoresed and analyzed. We classified MSI status as MSI-positive (instability at two or more microsatellite marker), and MSI-negative (no instability or instability at one marker). The CIMP status was evaluated by the MethyLight assay of eight markers (CACNA1G, CDKN2A [p16], CRABP1, IGF2, MLH1, NEUROG1, RUNX3, and SOCS1) [38]. We classified CRCs into CIMPnegative (0–4 methylated markers), CIMP-positive 1 (5–6 methylated markers), and CIMP-positive 2 (CIMP-P2) (7–8 methylated markers), as previously described [34].
- Statistical analyses
- SAS ver. 9.4 (SAS Institute Inc., Cary, NC, USA) and R software (http://www.r-project.org) were used for statistical analyses. Clinicopathological characteristics were compared between the three SMAD4 expression groups by use of chi-square test or Fisher exact test, as appropriate. Chi-square test for trend was used to compute p for trend. Survival curves after surgery were estimated with the Kaplan-Meier method, and differences in survival curves were tested with the log-rank test. All statistical tests were two-sided, and statistical significance was defined as p<.05.
- Ethics statement
- This study was approved by the Institutional Review Board which waived the requirement to obtain informed consent (IRB No. C-1502-029-647).
MATERIALS AND METHODS
- Clinicopathological and molecular correlation of nuclear SMAD4 expression in CRCs
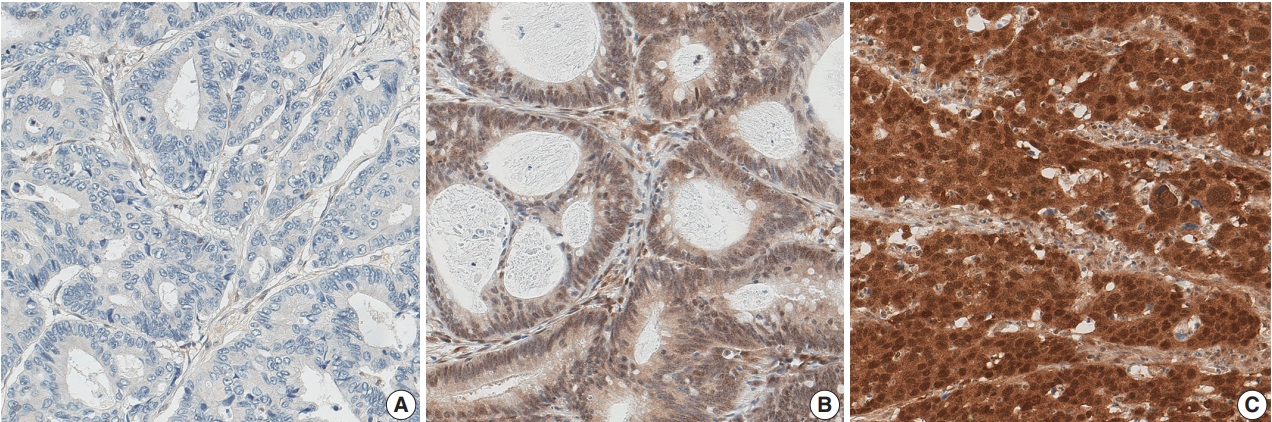
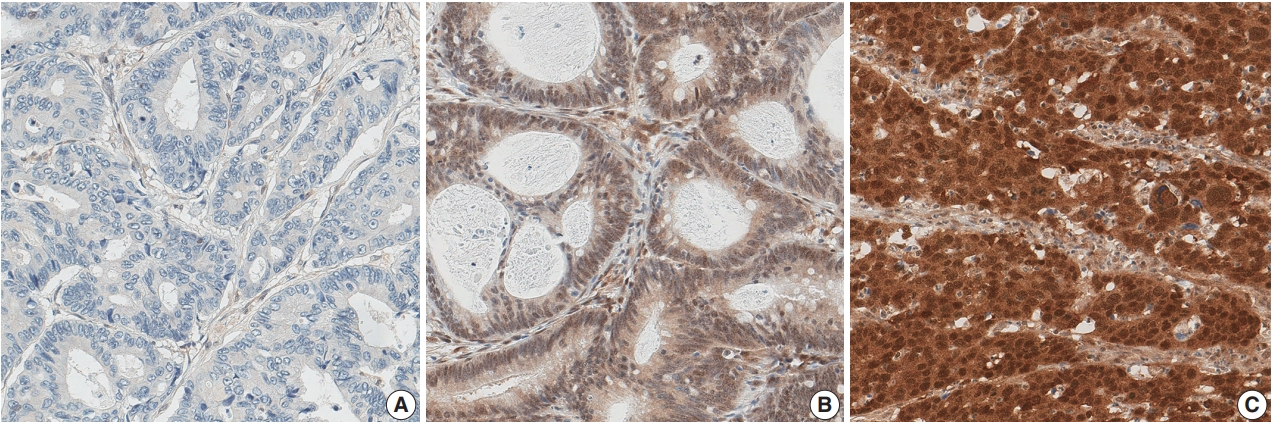
- Out of 1,281 CRC cases, 210 (16.4%) showed a loss of nuclear expression of SMAD4 (Fig. 1A). Among the remaining cases with retained SMAD4 expression, 942 cases (73.5%) showed low-level expression (Fig. 1B), while high-level expression was observed in 129 cases (10.1%) (Fig. 1C).
- We then sought to identify clinicopathologic features showing gradual changes according to the loss or overexpression of SMAD4 (Table 1). With decreasing expression of SMAD4, tumors tended to show an infiltrative gross type, more frequent lymphovascular and perineural invasion, tumor budding, and lower TILs (all p<.001). Consequently, CRC cases showed significant associations with higher pT (p<.001) and pN (p<.001) category, and frequent distant metastasis (p=.001) as nuclear SMAD4 expression decreased. Notably, statistically significant increase of SMAD4 loss and concomitant decrease of SMAD4 expression was noted as TNM stage increased (p <.001) (Supplementary Fig. S1).
- To identify molecular phenotypical correlates of such linear trends, we evaluated MSI, CIMP status, KRAS exon 2, BRAF codon 600, and immunohistochemical expression of CK7, CK20, and CDX2 (Table 2). With increasing expression of SMAD4, tumors tended to be more associated with MSI (p < .001), CIMP-P2 (p=.001), and MLH1 promoter methylation (p< .001). Interestingly, the expression of CK20 and CDX2 showed opposite trends with increasing SMAD2 expression; CDX2 expression was lost as nuclear SMAD4 expression increased (p= .001), while the expression of CK20 increased (p<.001).
- Prognostic implication of SMAD4 loss in CRCs
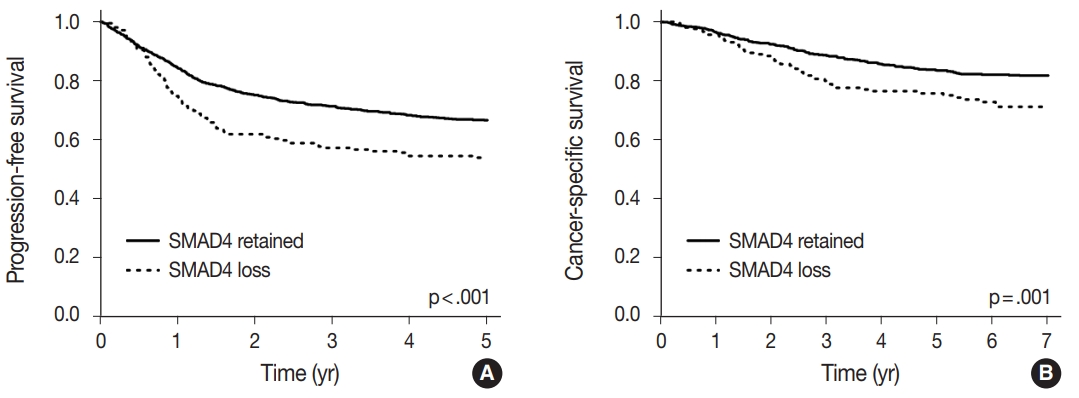
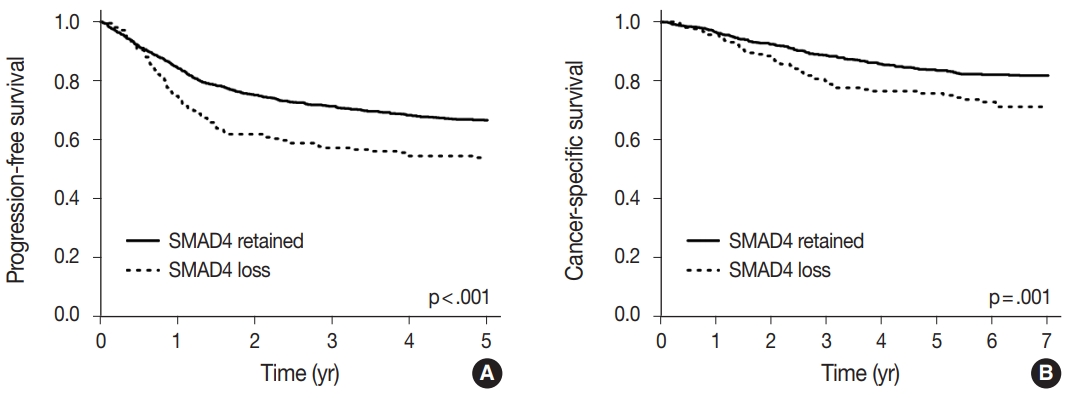
- A univariate survival analysis revealed that CRCs with a loss of nuclear SMAD4 expression exhibited a significantly worse 5-year progression-free survival (PFS) (p<.001) (Fig. 2A), and 7-year CSS (p=.001) (Fig. 2B). To explore whether there exists a differential prognostic effect of SMAD4 according to TNM stage, we performed the Kaplan-Meier analysis for each TNM stage subgroups (Supplementary Figs. S2, S3). In a stage-specific analysis, SMAD4 loss was associated with worse 5-year PFS (p = .019) in stage II CRCs, worse 5-year PFS (p = .055) and 7-year CSS (p=.011) in stage IV CRCs. In the multivariate Cox proportional hazard analysis, a loss of SMAD4 expression proved to be an independent prognostic factor for 5-year PFS (hazard ratio [HR], 1.27; 95% confidence interval [CI], 1.01 to 1.60; p=.042) (Table 3), and for 7-year CSS (HR, 1.45; 95% CI, 1.06 to 1.99; p=.022), after adjustment for TNM stage, tumor differentiation, adjuvant chemotherapy, and lymphovascular invasion.
RESULTS
- To the best of our knowledge, this is the second largest study focusing on the clinicopathologic and prognostic implications of SMAD4 expression in CRC, and the largest in an Asian population. While the largest previous study was performed on 1,381 stage II or III CRC patients enrolled at a pan-European clinical trial for adjuvant chemotherapy [12], our cohort consisted of 1,281 retrospectively collected patients ranging from stage I to IV. By combining CIMP analysis with additional histologic features such as tumor budding and TILs, we identified the general expression status of SMAD4 and its association with clinicopathologic and molecular features of CRC.
- Most previous studies evaluated SMAD4 expression based on two-tier classification: loss versus no loss, or low versus high expression [7,8,10,11,13-15,17,18,20]. Because of ambiguities in the definition of low expression, the reported prevalence of low-level SMAD4 expression varied significantly from 2.34% to 75.2%. On the other hand, studies that adopted a multi-tier classification reported relatively homogeneous results, with the prevalence of low-level SMAD4 expression ranging from 9.3% to 37.7% [6,9,12,16]. Considering the presence of an apparent loss, overexpression, and intermediate expression of SMAD4, we felt it more appropriate to use a three-tier classification (loss, low-level, and high-level) of SMAD4 expression. As a result, 16.4% of our cases were identified as SMAD4 loss, which falls within the range of previous reports.
- Consistent with the previous studies [8-17], we observed a significant association between SMAD4 loss and higher pT and pN category, and frequent distant metastasis. Consequently, we confirmed the value of SMAD4 loss as a prognostic factor for poor CSS and PFS of CRC. In colonic epithelial cells, TGF-β signaling reduces proliferation and promotes apoptosis and differentiation [39]. Because SMAD4 translocates to the nucleus by forming a heterodimeric complex with SMAD2/3 that is phosphorylated by the activated TGF-β receptor [2], it is intuitive that the immunohistochemical loss of nuclear SMAD4 expression suggests a concomitant loss of TGF-β signaling, which leads to uncontrolled proliferative behavior.
- Interestingly, we observed that some tumors overexpressed SMAD4 and those tumors tend to be MSI, CIMP-P2, and have their MLH1 promoter methylated. Along with the association between fungating gross type, these data collectively suggested an association between the sporadic MSI-high (MSI-H)/CIMP-high phenotype and overexpression of nuclear SMAD4. Although the meaning of SMAD4 overexpression is not straightforward, such a trend has been reported previously [9,12,40]. One possibility is that overexpression of SMAD4 in MSI-H tumors might be the consequence of a compensatory mechanism for mutational inactivation of the TGF-β signaling pathway; i.e., some machineries in the pathway, such as transforming growth factor β receptor II (TGFBR2) or activin type II receptor (ACVR2), are prone to mutations when mismatch repair is impaired [39]. At the same time, there are reports suggesting various bypass mechanisms to overcome such mutations [41-44]. Further mechanistic studies are needed to confirm this hypothesis.
- A novel finding in our study was the gradual increase of CDX2 expression as nuclear SMAD4 increased, while CK20 showed the opposite trend. Although this is in contrast with our previous report that expression of both CK20 and CDX2 got lost according to CpG island methylation [34], the proportional relationship of CDX2 and SMAD4 is consistent with some previous reports. In stomach, it has been reported that SMAD4 can activate the promoter of CDX2, and knockdown of SMAD4 led to the decreased expression of CDX2 [45]. Concomitant loss of SMAD4 and CDX2 was also observed in colorectal juvenile polyps obtained from juvenile polyposis syndrome patients [46]. The loss of CK20 expression could be explained as a consequence of epithelial mesenchymal transition (EMT). It is known that switch of intermediate filaments from cytokeratin to vimentin occurs during EMT [47]. It has been reported that the immunohistochemical expression of SMAD4 was positively correlated with that of EMT-related transcription factors such as Snail-1 and Twist-1 [48], and silencing of SMAD4 inhibited EMT [49]. Consequently, an intriguing hypothesis emerges that overexpression of SMAD4 accompanied loss of CK20 by promoting EMT.
- Consistent with a previous study [17], we observed a significant association between low TIL infiltration and SMAD4 loss. Although this could be a secondary effect of the MSI status, there are a series of reports on the correlation between SMAD4 loss and CCL15 expression [50-52]. These researchers demonstrated that the loss of SMAD4 upregulated CCL15, which resulted in recruitment of CCR1+ cells at the invasive front. CCR1+ cells are phenotypically myeloid-derived suppressor cells (MDSC) and express immunosuppressive molecules such as indoleamine 2,3- dioxygenase. Using mouse models, these researchers demonstrated that the loss of SMAD4 promoted pulmonary and hepatic metastasis through the CCL15-CCR1 axis. It is plausible that the MDSCs are also responsible for the lower TIL infiltration we observed for tumors with SMAD4 loss.
- In conclusion, we confirmed the value of determining expression of SMAD4 immunohistochemically as an independent prognostic factor for CRC in general. Furthermore, we identified some histologic and molecular features that might be clues to elucidate the role of SMAD4 in colorectal tumorigenesis and progression. Further studies are needed to validate these findings with an independent large-scale series of CRC cases.
DISCUSSION
Electronic Supplementary Material
-
Author contributions
Conceptualization: JMB, GHK.
Data curation: SYY, JMB.
Formal analysis: SYY, JMB.
Funding acquisition: JMB, GHK.
Investigation: SYY, JAL, JMB.
Methodology: SYY, JAL, YS, NYC, JMB.
Project administration: JMB, GHK.
Resources: JMB, GHK.
Supervision: JMB, GHK.
Validation: JMB, GHK.
Visualization: SYY, JMB.
Writing—original draft: SYY, JMB.
Writing—review & editing: JMB, GHK.
-
Conflicts of Interest
The authors declare that they have no potential conflicts of interest.
-
Funding
This study was supported by grants from the National Research Foundation (NRF) funded by the Korean Ministry of Science, ICT and Future Planning (2011-0030049, 2016M3 A9B6026921), Korean Health Technology R&D Project, Ministry of Health and Welfare (HI14C1277), and a grant from the SNUH Research Fund (04-2018-0620).
Notes
| SMAD4-loss (n = 210, 16.4%) | SMAD4-low (n = 942, 73.5%) | SMAD4-high (n = 129, 10.1%) | p for difference | p for trend | |
|---|---|---|---|---|---|
| KRAS mutation | .866 | .606 | |||
| Absent | 143 (68.1) | 657 (69.7) | 91 (70.5) | ||
| Present | 67 (31.9) | 285 (30.3) | 38 (29.5) | ||
| BRAF mutation (n = 1,278) | .094 | .330 | |||
| Absent | 196 (93.3) | 906 (96.5) | 122 (94.6) | ||
| Present | 14 (6.7) | 33 (3.5) | 7 (5.4) | ||
| MSI | < .001 | < .001 | |||
| MSS | 204 (97.1) | 889 (94.4) | 93 (72.1) | ||
| MSI | 6 (2.9) | 53 (5.6) | 36 (27.9) | ||
| CIMP | .229a | .001 | |||
| CIMP-N, P1 | 209 (99.5) | 930 (98.7) | 122 (94.6) | ||
| CIMP-P2 | 1 (0.5) | 12 (1.3) | 7 (5.4) | ||
| MLH1 promoter methylation | < .001 | < .001 | |||
| Unmethylated | 205 (97.6) | 921 (97.8) | 112 (86.8) | ||
| Methylated | 5 (2.4) | 21 (2.2) | 17 (13.2) | ||
| CK7 expression (n = 1,277) | .273 | .120 | |||
| Absent | 186 (89.4) | 868 (92.3) | 121 (93.8) | ||
| Present | 22 (10.6) | 72 (7.7) | 8 (6.2) | ||
| CK20 expression (n = 1,271) | < .001 | < .001 | |||
| Retained | 183 (88.8) | 780 (83.3) | 90 (69.8) | ||
| Loss | 23 (11.2) | 156 (16.7) | 39 (30.2) | ||
| CDX2 expression (n = 1,262) | .001 | .001 | |||
| Retained | 166 (80.6) | 831 (89.4) | 116 (91.3) | ||
| Loss | 40 (19.4) | 98 (10.6) | 11 (8.7) |
- 1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2018; 68: 394–424. ArticlePubMed
- 2. Zhao M, Mishra L, Deng CX. The role of TGF-beta/SMAD4 signaling in cancer. Int J Biol Sci 2018; 14: 111–23. ArticlePubMedPMC
- 3. Schutte M. DPC4/SMAD4 gene alterations in human cancer, and their functional implications. Ann Oncol 1999; 10 Suppl 4: 56–9. ArticlePubMed
- 4. Du Y, Zhou X, Huang Z, et al. Meta-analysis of the prognostic value of smad4 immunohistochemistry in various cancers. PLoS One 2014; 9: e110182. ArticlePubMedPMC
- 5. Wang JD, Jin K, Chen XY, Lv JQ, Ji KW. Clinicopathological significance of SMAD4 loss in pancreatic ductal adenocarcinomas: a systematic review and meta-analysis. Oncotarget 2017; 8: 16704–11. ArticlePubMed
- 6. Kouvidou C, Latoufis C, Lianou E, et al. Expression of Smad4 and TGF-beta2 in colorectal carcinoma. Anticancer Res 2006; 26: 2901–7. PubMed
- 7. Xu WQ, Jiang XC, Zheng L, Yu YY, Tang JM. Expression of TGF-beta1, TbetaRII and Smad4 in colorectal carcinoma. Exp Mol Pathol 2007; 82: 284–91. ArticlePubMed
- 8. Bacman D, Merkel S, Croner R, Papadopoulos T, Brueckl W, Dimmler A. TGF-beta receptor 2 downregulation in tumour-associated stroma worsens prognosis and high-grade tumours show more tumour-associated macrophages and lower TGF-beta1 expression in colon carcinoma: a retrospective study. BMC Cancer 2007; 7: 156.ArticlePubMedPMCPDF
- 9. Isaksson-Mettaväinio M, Palmqvist R, Dahlin AM, et al. High SMAD4 levels appear in microsatellite instability and hypermethylated colon cancers, and indicate a better prognosis. Int J Cancer 2012; 131: 779–88. ArticlePubMed
- 10. Voorneveld PW, Jacobs RJ, De Miranda NF, et al. Evaluation of the prognostic value of pSMAD immunohistochemistry in colorectal cancer. Eur J Cancer Prev 2013; 22: 420–4. ArticlePubMed
- 11. Kozak MM, von Eyben R, Pai J, et al. Smad4 inactivation predicts for worse prognosis and response to fluorouracil-based treatment in colorectal cancer. J Clin Pathol 2015; 68: 341–5. ArticlePubMed
- 12. Yan P, Klingbiel D, Saridaki Z, et al. Reduced expression of SMAD4 is associated with poor survival in colon cancer. Clin Cancer Res 2016; 22: 3037–47. ArticlePubMed
- 13. Jain S, Singhal S, Francis F, et al. Association of overexpression of TIF1gamma with colorectal carcinogenesis and advanced colorectal adenocarcinoma. World J Gastroenterol 2011; 17: 3994–4000. ArticlePubMedPMC
- 14. Handra-Luca A, Olschwang S, Fléjou JF. SMAD4 protein expression and cell proliferation in colorectal adenocarcinomas. Virchows Arch 2011; 459: 511–9. ArticlePubMedPDF
- 15. Chung Y, Wi YC, Kim Y, et al. The Smad4/PTEN expression pattern predicts clinical outcomes in colorectal adenocarcinoma. J Pathol Transl Med 2018; 52: 37–44. ArticlePubMedPDF
- 16. Oyanagi H, Shimada Y, Nagahashi M, et al. SMAD4 alteration associates with invasive-front pathological markers and poor prognosis in colorectal cancer. Histopathology 2019; 74: 873–82. ArticlePubMedPMC
- 17. Wasserman I, Lee LH, Ogino S, et al. SMAD4 loss in colorectal cancer patients correlates with recurrence, loss of immune infiltrate, and chemoresistance. Clin Cancer Res 2019; 25: 1948–56. ArticlePubMed
- 18. Alhopuro P, Alazzouzi H, Sammalkorpi H, et al. SMAD4 levels and response to 5-fluorouracil in colorectal cancer. Clin Cancer Res 2005; 11: 6311–6. ArticlePubMed
- 19. Boulay JL, Mild G, Lowy A, et al. SMAD4 is a predictive marker for 5-fluorouracil-based chemotherapy in patients with colorectal cancer. Br J Cancer 2002; 87: 630–4. ArticlePubMedPMCPDF
- 20. Alazzouzi H, Alhopuro P, Salovaara R, et al. SMAD4 as a prognostic marker in colorectal cancer. Clin Cancer Res 2005; 11: 2606–11. ArticlePubMed
- 21. Isaksson-Mettavainio M, Palmqvist R, Forssell J, Stenling R, Oberg A. SMAD4/DPC4 expression and prognosis in human colorectal cancer. Anticancer Res 2006; 26: 507–10. PubMed
- 22. Mesker WE, Liefers GJ, Junggeburt JM, et al. Presence of a high amount of stroma and downregulation of SMAD4 predict for worse survival for stage I-II colon cancer patients. Cell Oncol 2009; 31: 169–78. ArticlePubMedPMCPDF
- 23. Baraniskin A, Munding J, Schulmann K, et al. Prognostic value of reduced SMAD4 expression in patients with metastatic colorectal cancer under oxaliplatin-containing chemotherapy: a translational study of the AIO colorectal study group. Clin Colorectal Cancer 2011; 10: 24–9. ArticlePubMed
- 24. Wang C, Zhou Y, Ruan R, Zheng M, Han W, Liao L. High expression of COUP-TF II cooperated with negative Smad4 expression predicts poor prognosis in patients with colorectal cancer. Int J Clin Exp Pathol 2015; 8: 7112–21. PubMedPMC
- 25. Coates RF, Gardner JA, Gao Y, et al. Significance of positive and inhibitory regulators in the TGF-beta signaling pathway in colorectal cancers. Hum Pathol 2017; 66: 34–9. ArticlePubMed
- 26. Xie W, Rimm DL, Lin Y, Shih WJ, Reiss M. Loss of Smad signaling in human colorectal cancer is associated with advanced disease and poor prognosis. Cancer J 2003; 9: 302–12. ArticlePubMed
- 27. Gulubova M, Manolova I, Ananiev J, Julianov A, Yovchev Y, Peeva K. Role of TGF-beta1, its receptor TGFbetaRII, and Smad proteins in the progression of colorectal cancer. Int J Colorectal Dis 2010; 25: 591–9. ArticlePubMedPDF
- 28. Chun HK, Jung KU, Choi YL, et al. Low expression of transforming growth factor beta-1 in cancer tissue predicts a poor prognosis for patients with stage III rectal cancers. Oncology 2014; 86: 159–69. ArticlePubMed
- 29. Voorneveld PW, Jacobs RJ, Kodach LL, Hardwick JC. A Meta-analysis of SMAD4 immunohistochemistry as a prognostic marker in colorectal cancer. Transl Oncol 2015; 8: 18–24. ArticlePubMedPMC
- 30. Zhang B, Zhang B, Chen X, et al. Loss of Smad4 in colorectal cancer induces resistance to 5-fluorouracil through activating Akt pathway. Br J Cancer 2014; 110: 946–57. ArticlePubMedPMCPDF
- 31. Voorneveld PW, Kodach LL, Jacobs RJ, et al. The BMP pathway either enhances or inhibits the Wnt pathway depending on the SMAD4 and p53 status in CRC. Br J Cancer 2015; 112: 122–30. ArticlePubMedPDF
- 32. Chow E, Macrae F. A review of juvenile polyposis syndrome. J Gastroenterol Hepatol 2005; 20: 1634–40. ArticlePubMed
- 33. Ahn BK, Jang SH, Paik SS, Lee KH. Smad4 may help to identify a subset of colorectal cancer patients with early recurrence after curative therapy. Hepatogastroenterology 2011; 58: 1933–6. ArticlePubMed
- 34. Bae JM, Kim JH, Kwak Y, et al. Distinct clinical outcomes of two CIMP-positive colorectal cancer subtypes based on a revised CIMP classification system. Br J Cancer 2017; 116: 1012–20. ArticlePubMedPMCPDF
- 35. Bankhead P, Loughrey MB, Fernández JA, et al. QuPath: open source software for digital pathology image analysis. Sci Rep 2017; 7: 16878.ArticlePubMedPMCPDF
- 36. Bae JM, Lee TH, Cho NY, Kim TY, Kang GH. Loss of CDX2 expression is associated with poor prognosis in colorectal cancer patients. World J Gastroenterol 2015; 21: 1457–67. ArticlePubMedPMC
- 37. Boland CR, Thibodeau SN, Hamilton SR, et al. A National Cancer Institute Workshop on Microsatellite Instability for cancer detection and familial predisposition: development of international criteria for the determination of microsatellite instability in colorectal cancer. Cancer Res 1998; 58: 5248–57. PubMed
- 38. Weisenberger DJ, Siegmund KD, Campan M, et al. CpG island methylator phenotype underlies sporadic microsatellite instability and is tightly associated with BRAF mutation in colorectal cancer. Nat Genet 2006; 38: 787–93. ArticlePubMedPDF
- 39. Jung B, Staudacher JJ, Beauchamp D. Transforming growth factor beta superfamily signaling in development of colorectal cancer. Gastroenterology 2017; 152: 36–52. ArticlePubMed
- 40. Salovaara R, Roth S, Loukola A, et al. Frequent loss of SMAD4/DPC4 protein in colorectal cancers. Gut 2002; 51: 56–9. ArticlePubMedPMC
- 41. Ilyas M, Efstathiou JA, Straub J, Kim HC, Bodmer WF. Transforming growth factor beta stimulation of colorectal cancer cell lines: type II receptor bypass and changes in adhesion molecule expression. Proc Natl Acad Sci U S A 1999; 96: 3087–91. ArticlePubMedPMC
- 42. Deacu E, Mori Y, Sato F, et al. Activin type II receptor restoration in ACVR2-deficient colon cancer cells induces transforming growth factor-beta response pathway genes. Cancer Res 2004; 64: 7690–6. ArticlePubMed
- 43. Baker K, Raut P, Jass JR. Microsatellite unstable colorectal cancer cell lines with truncating TGFbetaRII mutations remain sensitive to endogenous TGFbeta. J Pathol 2007; 213: 257–65. ArticlePubMed
- 44. de Miranda NF, van Dinther M, van den Akker BE, van Wezel T, ten Dijke P, Morreau H. Transforming growth factor beta signaling in colorectal cancer cells with microsatellite instability despite biallelic mutations in TGFBR2. Gastroenterology 2015; 148: 1427–37. e8. ArticlePubMed
- 45. Barros R, Pereira B, Duluc I, et al. Key elements of the BMP/SMAD pathway co-localize with CDX2 in intestinal metaplasia and regulate CDX2 expression in human gastric cell lines. J Pathol 2008; 215: 411–20. ArticlePubMed
- 46. Barros R, Mendes N, Howe JR, et al. Juvenile polyps have gastric differentiation with MUC5AC expression and downregulation of CDX2 and SMAD4. Histochem Cell Biol 2009; 131: 765–72. ArticlePubMedPDF
- 47. Savagner P. The epithelial-mesenchymal transition (EMT) phenomenon. Ann Oncol 2010; 21 Suppl 7: vii89–92. ArticlePubMedPMCPDF
- 48. Ioannou M, Kouvaras E, Papamichali R, Samara M, Chiotoglou I, Koukoulis G. Smad4 and epithelial-mesenchymal transition proteins in colorectal carcinoma: an immunohistochemical study. J Mol Histol 2018; 49: 235–44. ArticlePubMedPDF
- 49. Deckers M, van Dinther M, Buijs J, et al. The tumor suppressor Smad4 is required for transforming growth factor beta-induced epithelial to mesenchymal transition and bone metastasis of breast cancer cells. Cancer Res 2006; 66: 2202–9. ArticlePubMed
- 50. Itatani Y, Kawada K, Fujishita T, et al. Loss of SMAD4 from colorectal cancer cells promotes CCL15 expression to recruit CCR1+ myeloid cells and facilitate liver metastasis. Gastroenterology 2013; 145: 1064–75. e11. ArticlePubMed
- 51. Inamoto S, Itatani Y, Yamamoto T, et al. Loss of SMAD4 promotes colorectal cancer progression by accumulation of myeloid-derived suppressor cells through the CCL15-CCR1 chemokine axis. Clin Cancer Res 2016; 22: 492–501. ArticlePubMed
- 52. Yamamoto T, Kawada K, Itatani Y, et al. Loss of SMAD4 promotes lung metastasis of colorectal cancer by accumulation of CCR1+ tumor-associated neutrophils through CCL15-CCR1 axis. Clin Cancer Res 2017; 23: 833–44. ArticlePubMed
References
Figure & Data
References
Citations

- Unraveling Resistance to Immunotherapy in MSI-High Colorectal Cancer
Ronald Heregger, Florian Huemer, Markus Steiner, Alejandra Gonzalez-Martinez, Richard Greil, Lukas Weiss
Cancers.2023; 15(20): 5090. CrossRef - Association of β-Catenin, APC, SMAD3/4, Tp53, and Cyclin D1 Genes in Colorectal Cancer: A Systematic Review and Meta-Analysis
Hongfeng Yan, Fuquan Jiang, Jianwu Yang, Ying-Kun Xu
Genetics Research.2022; 2022: 1. CrossRef - Comprehensive genetic features of gastric mixed adenoneuroendocrine carcinomas and pure neuroendocrine carcinomas
Jiwon Koh, Soo Kyung Nam, Yoonjin Kwak, Gilhyang Kim, Ka‐Kyung Kim, Byung‐Chul Lee, Sang‐Hoon Ahn, Do Joong Park, Hyung‐Ho Kim, Kyoung Un Park, Woo Ho Kim, Hye Seung Lee
The Journal of Pathology.2021; 253(1): 94. CrossRef - Alterations of PTEN and SMAD4 methylation in diagnosis of breast cancer: implications of methyl II PCR assay
Menha Swellam, Entsar A. Saad, Shimaa Sabry, Adel Denewer, Camelia Abdel Malak, Amr Abouzid
Journal of Genetic Engineering and Biotechnology.2021; 19(1): 54. CrossRef - Molecular Characterization and Functional Analysis of Two Steroidogenic Genes TSPO and SMAD4 in Yellow Catfish
Fang Chen, Chong-Chao Zhong, Chang-Chun Song, Shu-Wei Chen, Yang He, Xiao-Ying Tan
International Journal of Molecular Sciences.2021; 22(9): 4505. CrossRef - SMAD7 and SMAD4 expression in colorectal cancer progression and therapy response
Jovana Rosic, Sandra Dragicevic, Marko Miladinov, Jovana Despotovic, Aleksandar Bogdanovic, Zoran Krivokapic, Aleksandra Nikolic
Experimental and Molecular Pathology.2021; 123: 104714. CrossRef - Actionable Potentials of Less Frequently Mutated Genes in Colorectal Cancer and Their Roles in Precision Medicine
Ryia Illani Mohd Yunos, Nurul Syakima Ab Mutalib, Francis Yew Fu Tieng, Nadiah Abu, Rahman Jamal
Biomolecules.2020; 10(3): 476. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
- Related articles
-
- Clinicopathologic implication of PD-L1 gene alteration in primary adrenal diffuse large B cell lymphoma
- A comparative prognostic performance of definitions of Crohn-like lymphoid reaction in colorectal carcinoma
- Clinicopathologic characteristics of HER2-positive pure mucinous carcinoma of the breast

 E-submission
E-submission