Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 50(4); 2016 > Article
-
Brief Case Report
IgG4-Related Sclerosing Mesenteritis - Seok Joo Lee, Cheol Keun Park, Woo Ick Yang, Sang Kyum Kim
-
Journal of Pathology and Translational Medicine 2016;50(4):309-311.
DOI: https://doi.org/10.4132/jptm.2015.12.03
Published online: January 11, 2016
Department of Pathology, Yonsei University Medical Center, Seoul, Korea
- Corresponding Author Sang Kyum Kim, MD, PhD Department of Pathology, Yonsei University Medical Center, 50-1 Yonsei-ro, Seodaemun-gu, Seoul 03722, Korea Tel: +82-2-2228-6751, Fax: +82-2-2227-7939, E-mail: 'nicekyumi@yuhs.ac'
• Received: October 6, 2015 • Revised: November 27, 2015 • Accepted: December 3, 2015
© 2016 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
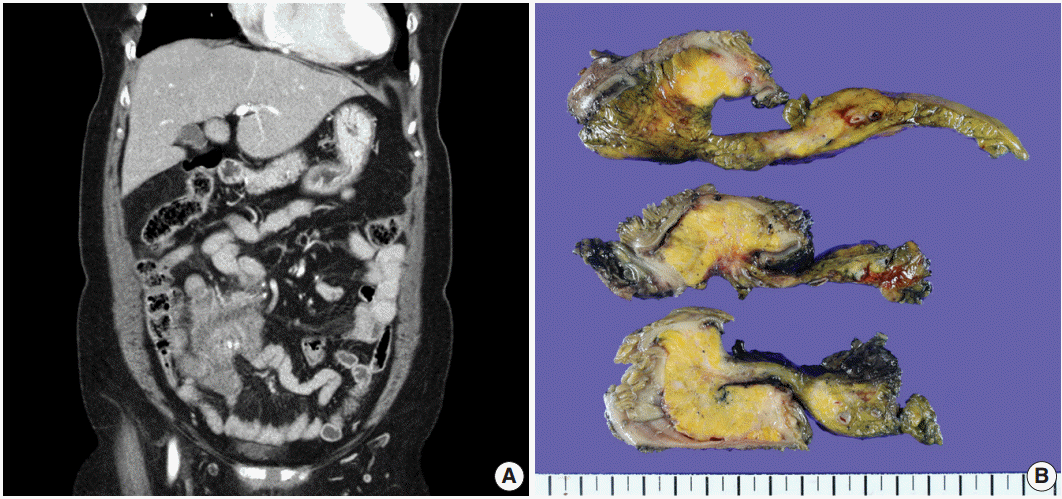
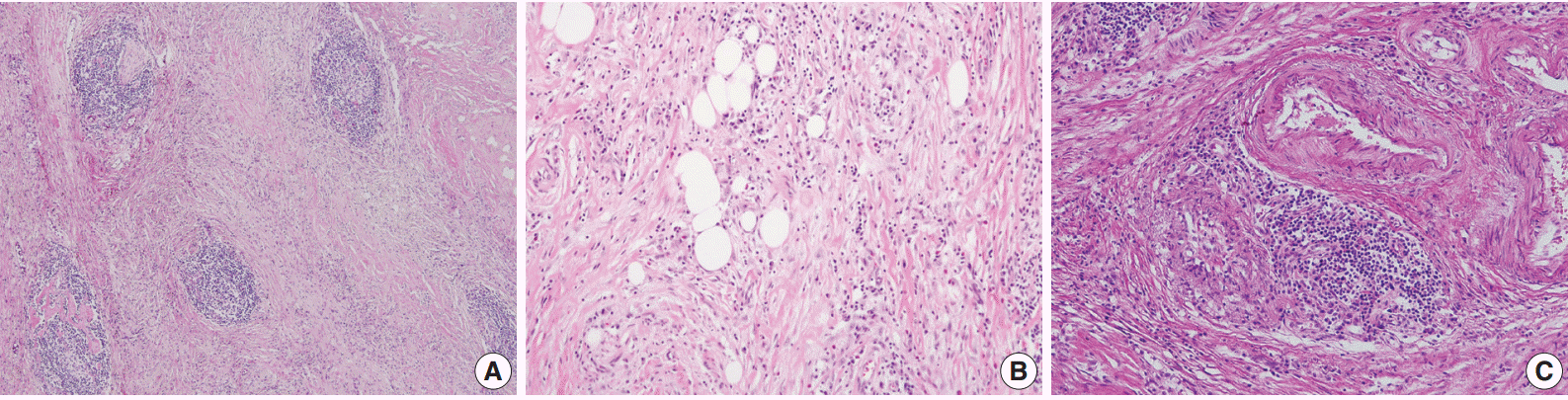
- A 70-year-old woman presented with a palpable intra-abdominal mass and pain for about 3 months. She had a history of hypertension and a total hysterectomy 10 years earlier. Radiologically, a 7.9 cm lobulated mass was found in her right lower abdomen with multiple calcifications (Fig. 1A), suggesting a mesenteric-origin tumor. Positron emission tomography–computed tomography revealed a mesenteric mass with calcification in the rihgt lower quadrant of abdomen which had intense fluorodeoxyglucose (FDG) uptake. A few satellite nodules were also present in the mesentery; however, no other abnormal FDG uptake was identified. The patient underwent segmental resection of the small bowel. Grossly, the mucosal surface was intact and a multilobulated mass with whitish fibrosis was noted in the mesentery (Fig. 1B). Light microscopy revealed fat necrosis and a sclerosing fibrotic lesion including lymphocytic aggregation with germinal center formation and spindle cell proliferation (Fig. 2A). The spindle cells had abundant cytoplasm, a vesicular nucleus, and prominent nucleoli (Fig. 2B). In the periphery, acute inflammatory cells and eosinophils were seen. Chronic inflammatory cell infiltration into the venular wall with obliteration of the lumen juxtaposed to an artery was also seen (Fig. 2C).
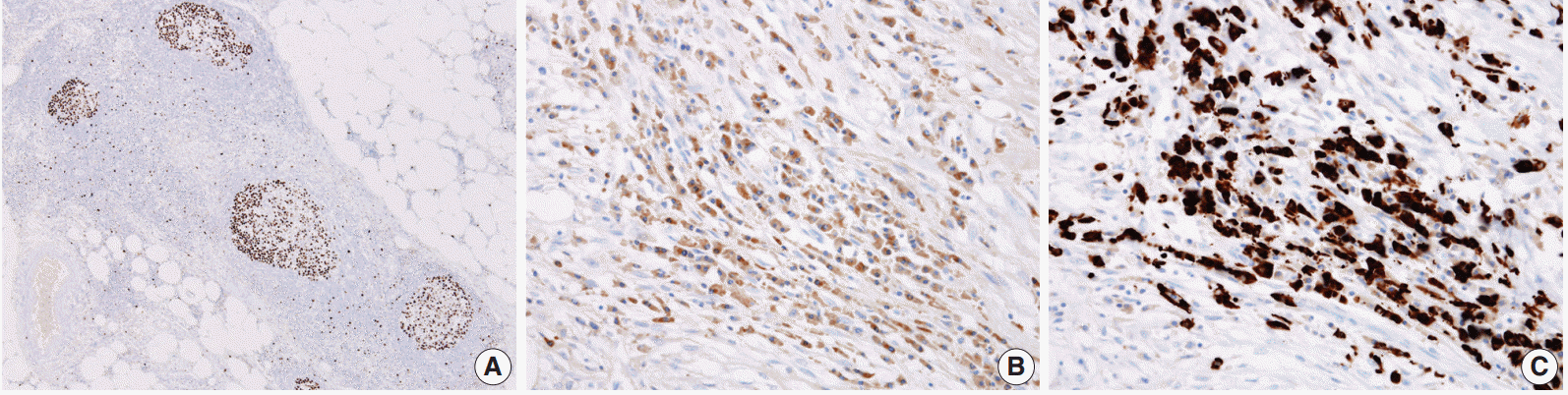
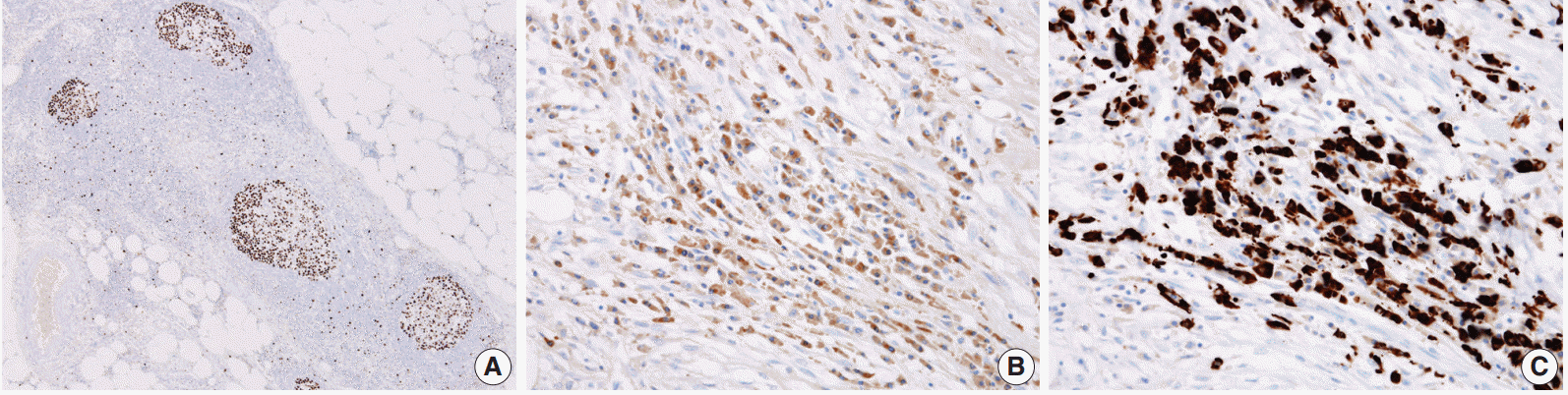
- As a result, three histologic findings of storiform pattern of fibrosis, lymphoplasmacytic infiltration, and obliterative phlebitis were distinct features of the presented case. Based on these findings, the differential diagnoses included inflammatory malignant fibrous histiocytoma, inflammatory myofibroblastic tumor, and SM. We performed immunohistochemical staining using anaplastic lymphoma kinase (ALK, D5F3), smooth muscle actin (SMA), Ki-67, IgG, and IgG4 antibodies. The overall Ki-67 labeling index was low and was increased only in lymphoid follicles (Fig. 3A). ALK staining was negative and SMA staining revealed vascular obliteration (data not shown). Most of the plasma cells expressed IgG (Fig. 3B) and IgG4 (Fig. 3C), and the IgG4-positive cells/IgG-positive cells ratio was higher than 90%. Therefore, we concluded the lesion as IgG4-RSM. Immediately after the surgery, blood samples were taken to measure the patient’s levels of IgG and IgG4. IgG4 level has greatly increased (2,130.0 mg/L; reference range, 39.2 to 864.0 mg/L), while IgG stayed within the normal range (1,318 mg/dL; reference range, 700 to 1,600 mg/dL). The patient was discharged without complication, received a clinical work-up for IgG4-RSM in an outpatient department, and started on steroid therapy.
CASE REPORT
- SM is a rare benign condition that presents with fibrosis, inflammation, and fat necrosis and occurs idiopathically in the small bowel mesentery [2]. Clinically, patients with SM mostly complain of chronic severe pain and chronic non-specific problems such as nausea, vomiting, diarrhea, cramping, weight loss, and fever [4]. Histologically, SM shows fibrosis with fat necrosis, chronic inflammation especially around the vessels, and variable focal calcification. In particular, if the immunologic and histologic characteristics are consistent with the IgG4-RD, it is classified as IgG4-RSM [3]. The histologic features of IgG4-RSM include more than two of the followings: lymphoplasmacytic infiltration, storiform fibrosis, and obliterative phlebitis. The immunologic criterion is IgG4-positive/IgG-positive cells > 40%, but this ratio is considered secondary in importance to the histological appearance.
- Kerdsirichairat et al. [3] conducted a systematic literature review of SM with IgG4 testing, and 11 out of 18 cases in total met the criteria for IgG4-RSM. The age of patients with IgG4-RSM ranges from 48 to 82. IgG4-RSM has a male predominance similar to SM. IgG4-RSM is known as a rare mesenteric disease of unknown etiology, although prior abdominal surgery can result in SM. Emory et al. [2] reviewed 84 cases of SM and a history of trauma or surgery was present in four of 84 patients. Two of four patients had hysterectomies 20 and 12 years earlier. Our patient has a history of total abdominal hysterectomy, which might be the cause of the IgG4-RSM.
- There is no generalized consensus regarding the treatment of SM, including medical therapy, surgical therapy, and surgery with additional medical therapy. In one study, the response rates were shown to be good for all therapies (65%, 82%, and 71%, respectively) [3], but the limited number of cases made it difficult to determine a treatment of choice. Even, when patients with SM are not treated, they show a high rate of spontaneous resolution [5]. Therefore, therapy is needed when a patient displays symptoms.
- In our case, the patient’s differential diagnoses included inflammatory malignant fibrous histiocytoma and inflammatory myofibroblastic tumor, based on microscopic features. Inflammatory malignant fibrous histiocytoma shows neoplastic histiocyte-like cells mixed with neutrophils and other inflammatory cells. It has a high Ki-67 index, but in our patient’s case, the overall Ki-67 index was low and was only increased in lymphoid follicles. Inflammatory myofibroblastic tumor shows spindle cell proliferation with myofibroblastic differentiation, a collagen stroma, and chronic inflammatory cell infiltrations. However, it rarely shows obliterative phlebitis [6]. It is immunoreactive for ALK in about half the cases and stains for SMA [7,8]. Also, the ratio of IgG4-positive/IgG-positive cells is markedly lower than that of IgG4-RD [9]. In our case, SMA staining was non-specific and only confirmed the obliterative phlebitis. The high ratio of IgG4-positive/IgG-positive cells also supported IgG4-RSM.
- In summary, we present a case of IgG4-RSM, which is compatible with previously described cases. The condition is benign and has a self-limiting course, but the pathologic diagnosis is important to exclude malignant conditions, which can be misdiagnosed on radiologic analysis. The rarity of the disease limits its clear characterization, and more cases and studies are needed.
DISCUSSION
-
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Notes
Fig. 1.(A) A 7.9 cm lobulated mass with multiple variable calcifications is seen in the right lower abdomen on abdominal computed tomography. (B) An ill-defined whitish fibrotic lesion is noted in the mesentery.
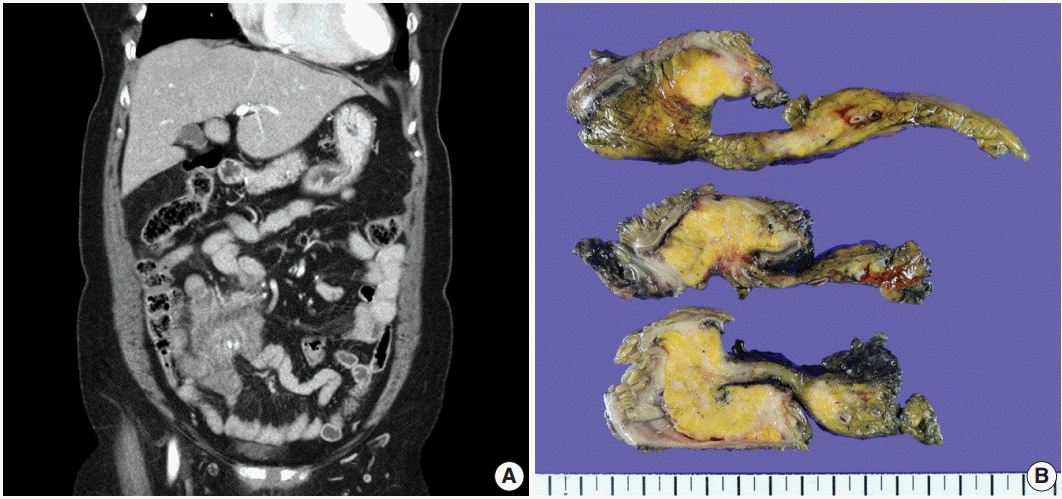

Fig. 2.(A, B) The microscopic findings show fat necrosis and a sclerosing fibrotic lesion (storiform fibrosis) with lymphocytic aggregation within the germinal center formation and spindle cell proliferation. (C) Obliterative phlebitis just next to an artery is also seen.
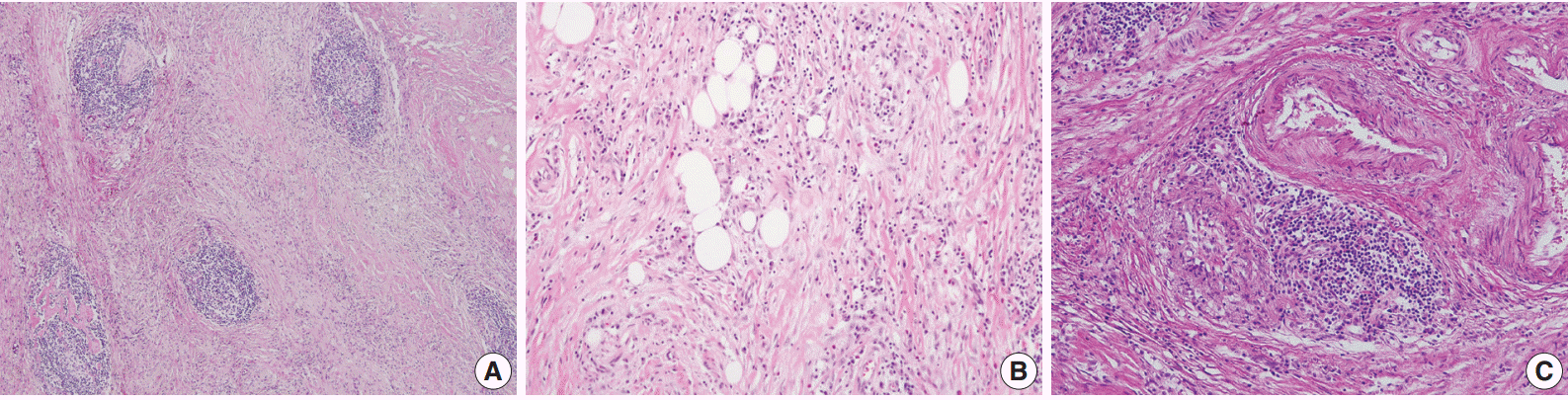

Fig. 3.(A) A low Ki-67–labeling index is shown with increase only in the lymphoid follicle. The IgG4-positive cells (C)/IgG-positive cells (B) ratio is higher than 90%.

- 1. Sulla JV. Mesenterite e sclerosante. Policlinico Prat 1924; 31: 575–81.
- 2. Emory TS, Monihan JM, Carr NJ, Sobin LH. Sclerosing mesenteritis, mesenteric panniculitis and mesenteric lipodystrophy: a single entity? Am J Surg Pathol 1997; 21: 392–8. ArticlePubMed
- 3. Kerdsirichairat T, Mesa H, Abraham J, et al. Sclerosing mesenteritis and IgG4-related mesenteritis: case series and a systematic review of natural history and response to treatments. Immunogastroenterology 2013; 2: 119–28. Article
- 4. Parra-Davila E, McKenney MG, Sleeman D, et al. Mesenteric panniculitis: case report and literature review. Am Surg 1998; 64: 768–71. PubMed
- 5. Akram S, Pardi DS, Schaffner JA, Smyrk TC. Sclerosing mesenteritis: clinical features, treatment, and outcome in ninety-two patients. Clin Gastroenterol Hepatol 2007; 5: 589–96. ArticlePubMed
- 6. Minato H, Shimizu J, Arano Y, et al. IgG4-related sclerosing mesenteritis: a rare mesenteric disease of unknown etiology. Pathol Int 2012; 62: 281–6. ArticlePubMed
- 7. Cessna MH, Zhou H, Sanger WG, et al. Expression of ALK1 and p80 in inflammatory myofibroblastic tumor and its mesenchymal mimics: a study of 135 cases. Mod Pathol 2002; 15: 931–8. ArticlePubMed
- 8. Qiu X, Montgomery E, Sun B. Inflammatory myofibroblastic tumor and low-grade myofibroblastic sarcoma: a comparative study of clinicopathologic features and further observations on the immunohistochemical profile of myofibroblasts. Hum Pathol 2008; 39: 846–56. ArticlePubMed
- 9. Yamamoto H, Yamaguchi H, Aishima S, et al. Inflammatory myofibroblastic tumor versus IgG4-related sclerosing disease and inflammatory pseudotumor: a comparative clinicopathologic study. Am J Surg Pathol 2009; 33: 1330–40. ArticlePubMed
References
Figure & Data
References
Citations
Citations to this article as recorded by 

- Manifestaciones toracoabdominales de las enfermedades relacionadas con la inmunoglobulina G4
M. Álvarez García, J.C. Álvarez Torices, M.J. Díez Liébana, M.R. de la Fuente Olmos
Radiología.2023; 65(2): 165. CrossRef - Thoracoabdominal manifestations of immunoglobulin G4-related disease
M. Álvarez García, J.C. Álvarez Torices, M.J. Díez Liébana, M.R. de la Fuente Olmos
Radiología (English Edition).2023; 65(2): 165. CrossRef - Systemic manifestations – do not forget the small bowel
Stefania Chetcuti Zammit, Reena Sidhu
Current Opinion in Gastroenterology.2021; 37(3): 234. CrossRef - A case of immunoglobulin G4-related sclerosing mesenteritis without other organ involvement
Masaya Kozono, Shiroh Tanoue, Kanna Kiyama, Kenichi Jikuya, Machiko Kawahira, Makoto Hinokuchi, Hiromichi Iwaya, Shiho Arima, Shinichi Hashimoto, Kiyokazu Hiwatashi, Yoshio Fukuda, Masahiko Sakoda, Michiyo Higashi, Koichi Tokushige, Akio Ido
Clinical Journal of Gastroenterology.2021; 14(5): 1411. CrossRef - Sclerosing Mesenteritis Mimicking IgG4-related Disease
Masahide Fukuda, Toru Miyake, Akiko Matsubara, Nobuyasu Ikai, Eri Tanaka, Tomo Namura, Yasuhiro Wada, Mai Noujima, Suzuko Moritani, Kazunari Murakami, Akira Andoh, Masaji Tani, Ryoji Kushima
Internal Medicine.2020; 59(4): 513. CrossRef - A rare case report of immunoglobulin G4-related sclerosing mesenteritis and review of the literature
Zhicheng Liu, Yan Jiao, Liang He, Helei Wang, Daguang Wang
Medicine.2020; 99(41): e22579. CrossRef - Calcifying fibrous tumor of the gastrointestinal tract: A clinicopathologic review and update
Donald Turbiville, Xu-Chen Zhang
World Journal of Gastroenterology.2020; 26(37): 5597. CrossRef - Immunoglobulin G4 Unrelated Idiopathic Mesenteric Sclerosis
Tae Hyung Kwon, Kwang Bum Cho, Hyun Jik Lee, Sun Young Kwon, Yoon Suk Lee
The Korean Journal of Gastroenterology.2019; 73(1): 50. CrossRef - A Case of IgG4-related Sclerosing Mesenteritis Causing Intestinal Perforation
Junich SAKAMOTO, Masahiko WATANABE
Nihon Rinsho Geka Gakkai Zasshi (Journal of Japan Surgical Association).2019; 80(9): 1658. CrossRef - „Misty mesentery“
L. P. Beyer, A. Schreyer
Der Radiologe.2018; 58(1): 56. CrossRef - IgG4-Related Disease Mimicking Crohn’s Disease: A Case Report and Review of Literature
Fabiana Ciccone, Antonio Ciccone, Mirko Di Ruscio, Filippo Vernia, Gianluca Cipolloni, Gino Coletti, Giuseppe Calvisi, Giuseppe Frieri, Giovanni Latella
Digestive Diseases and Sciences.2018; 63(4): 1072. CrossRef - Mesenteric tumour deposits arising from small‐intestine neuroendocrine tumours are frequently associated with fibrosis and IgG4‐expressing plasma cells
Jordan Roberts, Raul S Gonzalez, Frank Revetta, Chanjuan Shi
Histopathology.2018; 73(5): 795. CrossRef - A Case Report of Probable Diagnosis Group of IgG4-related Sclerosing Mesenteritis
Daiki YOKOYAMA, Takayuki TAJIMA, Masaya MUKAI, Shuji UDA, Shinichiroh HIRAIWA, Takuma TAJIRI, Eiji NOMURA, Hiroyasu MAKUUCHI
Nihon Rinsho Geka Gakkai Zasshi (Journal of Japan Surgical Association).2017; 78(10): 2245. CrossRef - Serosal involvement in IgG4-related disease: report of two cases and review of the literature
Juan González-Moreno, Inés Losada-López, Cristina Gállego-Lezaun, Mercedes García-Gasalla, Cristina Gómez Bellvert, Norberto Ortego Centeno
Rheumatology International.2016; 36(7): 1033. CrossRef - IgG4-related sclerosing mesenteritis causing bowel obstruction: a case report
Atsushi Abe, Tatsuya Manabe, Nobuyoshi Takizawa, Takashi Ueki, Daisuke Yamada, Kinuko Nagayoshi, Yoshihiko Sadakari, Hayato Fujita, Shuntaro Nagai, Hidetaka Yamamoto, Yoshinao Oda, Masafumi Nakamura
Surgical Case Reports.2016;[Epub] CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure

 E-submission
E-submission