Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(5); 2013 > Article
-
Original Article
ALK-Positive Renal Cell Carcinoma in a Large Series of Consecutively Resected Korean Renal Cell Carcinoma Patients - Cheol Lee1, Jeong Whan Park1, Ja Hee Suh1, Kyung Han Nam1, Kyung Chul Moon1,2
-
Korean Journal of Pathology 2013;47(5):452-457.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.5.452
Published online: October 25, 2013
1Department of Pathology, Seoul National University College of Medicine, Seoul, Korea.
2Kidney Research Institute, Medical Research Center, Seoul National University College of Medicine, Seoul, Korea.
- Corresponding Author: Kyung Chul Moon, M.D. Department of Pathology, Kidney Research Institute, Medical Research Center, Seoul National University College of Medicine, 103 Daehak-ro, Jongno-gu, Seoul 110-799, Korea. Tel: +82-2-2072-1767, Fax: +82-2-743-5530, 'blue7270@snu.ac.kr'
• Received: September 13, 2013 • Revised: September 30, 2013 • Accepted: October 1, 2013
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Recently, there have been a few reports of renal cell carcinoma (RCC) cases with anaplastic lymphoma kinase (ALK) gene fusion. In this study, we screened consecutively resected RCCs from a single institution for ALK protein expression by immunohistochemistry, and then we performed fluorescence in situ hybridization to confirm the ALK gene alteration in ALK immunohistochemistry-positive cases.
-
Methods
- We screened 829 RCCs by ALK immunohistochemistry, and performed fluorescence in situ hybridization analysis using ALK dual-color break-apart rearrangement probe. Histological review and additional immunohistochemistry analyses were done in positive cases.
-
Results
- One ALK-positive case was found. Initial diagnosis of this case was papillary RCC type 2. This comprises 0.12% of all RCCs (1/829) and 1.9% of papillary RCCs (1/53). This patient was a 44-year-old male with RCC found during routine health check-up. He was alive without evidence of disease 12 years after surgery. The tumor showed a papillary and tubular pattern, and showed positivity for CD10 (focal), epithelial membrane antigen, cytokeratin 7, pan-cytokeratin, PAX-2, and vimentin.
-
Conclusions
- We found the first RCC case with ALK gene rearrangement in Korean patients by ALK immunohistochemistry among 829 RCCs. This case showed similar histological and immunohistochemical features to those of previous adult cases with ALK rearrangement, and showed relatively good prognosis.
- Patients and tissue microarray
- We examined 829 RCCs from patients who underwent radical or partial nephrectomy between 1995 and 2005 at the Seoul National University Hospital. RCCs included 689 (83.1%) clear cell RCC, 73 (8.8%) chromophobe RCC, 53 (6.4%) papillary RCC, and 14 (1.7%) other rare subtypes. Tissue microarray (TMA) blocks consisted of a representative tumor core section (2 mm in diameter) from each formalin-fixed paraffin block (SuperBioChips Laboratories, Seoul, Korea). Clinical and pathological information were collected from electronic medical records and pathologic reports. This study was approved by the Institutional Review Board (IRB) of Seoul National University Hospital.
- Immunohistochemistry
- Immunohistochemical staining for ALK expression was performed on 4 µm-thick sections taken from the TMA blocks. Monoclonal mouse anti-human ALK antibody (Novocastra, Newcastle upon Tyne, UK) was diluted 1:100 and immunohistochemistry was performed using the Ventana Benchmark XT automated staining system (Ventana Medical Systems, Tucson, AZ, USA).15-17 Additional immunohistochemistry for CD10 (ready-to-use, Novocastra,), cytokeratin 7 (1:300, Dako, Glostrup, Denmark), cytokeratin 20 (1:50, Dako), pan-cytokeratin (1:300, Dako), PAX-2 (1:100, Epitomics, Burlingame, CA, USA), epithelial membrane antigen (EMA; 1:300, Dako), vimentin (1:500, Dako), alpha-methylacyl-CoA racemase (AMACR; 1:300, Dako), c-kit (1:300, Dako), TFE3 (1:1,500, Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA), and human melanoma black 45 (HMB45; 1:200, Dako) were also performed for ALK immunohistochemistry-positive cases.
- Fluorescence in situ hybridization analysis
- For analysis of the ALK gene alteration, we performed FISH analysis for ALK immunohistochemistry-positive cases. Two-micrometer-thick sections were deparaffinized, dehydrated, immersed in 0.2 N HCl, boiled in a microwave in citrate buffer (pH 6.0), and incubated in 1 M NaSCN for 35 minutes at 80℃. Sections were then immersed in pepsin solution and the tissues were fixed in 10% neutral-buffered formalin. Dual-probe hybridization was performed as described previously22 using Vysis ALK dual-color break-apart rearrangement probe (Abbott Molecular, Abbott Park, IL, USA). FISH signals for each locus-specific FISH probe were assessed under an Olympus BX51TRF microscope (Olympus, Tokyo, Japan) equipped with a triple-pass filter (DAPI/Green/Orange).
MATERIALS AND METHODS
- Identification of ALK immunohistochemical positivity and ALK gene alteration
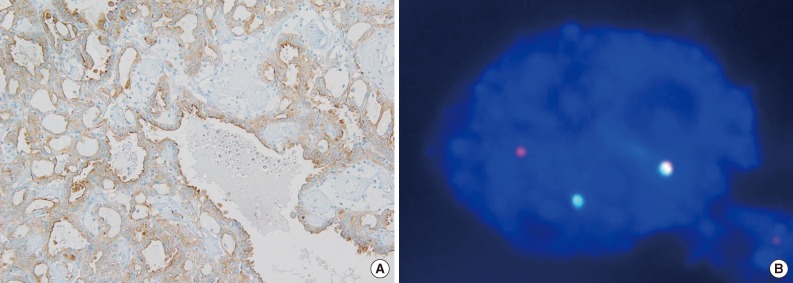
- ALK immunohistochemical staining showed only one positive case among 829 RCCs (Fig. 1A), and there were no cases showing borderline positivity. The initial diagnosis of this case was papillary RCC type 2. This case also revealed the break-apart signals in ALK FISH analysis (Fig. 1B). This comprises of 0.12% of all RCCs (1/829) and 1.9% of papillary RCCs (1/53).
- Patient information
- The patient was a 44-year-old male. The RCC was incidentally found by ultrasonography during a routine health check-up and radical nephrectomy was done. The patient had no history of sickle cell trait. He was alive, without evidence of disease, 12 years after surgery.
- Pathologic findings
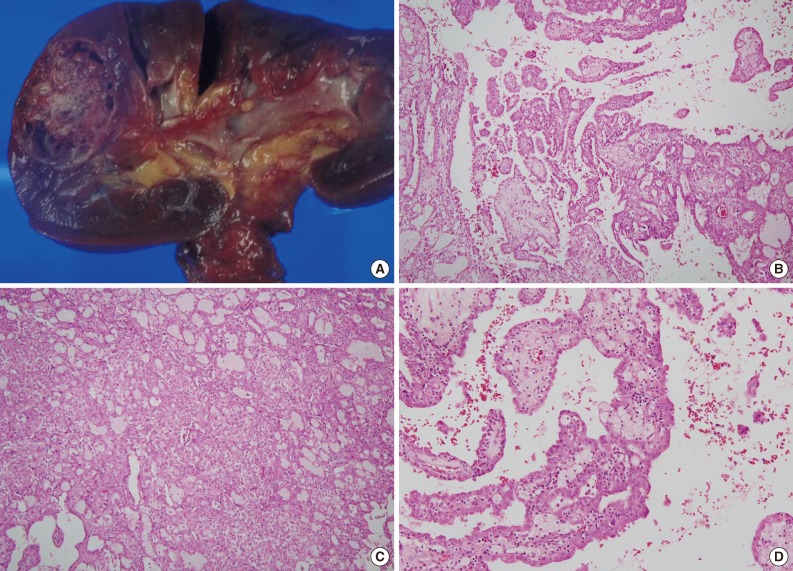
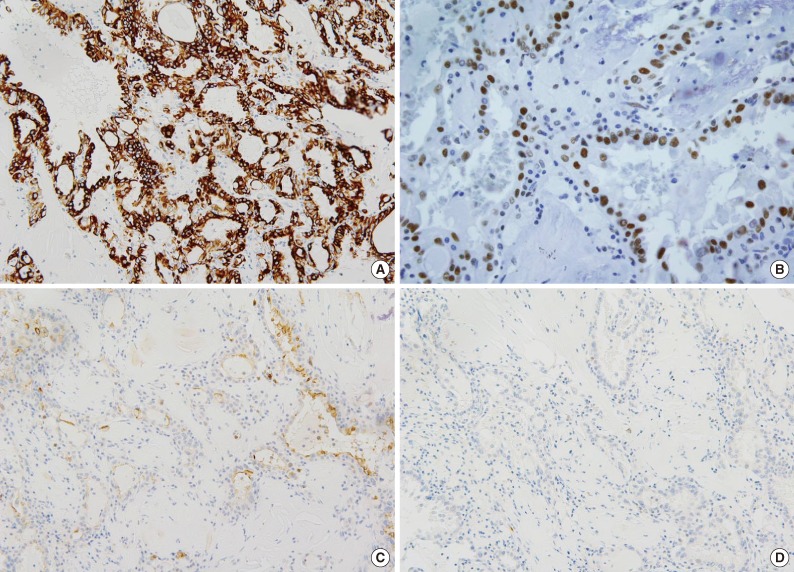
- The size of tumor was 3×3×2.5 cm. The tumor was confined to the renal parenchyme and there was no regional lymph node metastases in three regional lymph nodes (pT1aN0 by American Joint Committee on Cancer [AJCC] 7th edition23). Grossly, the tumor was a well-circumscribed, partially cystic mass (Fig. 2A). Microscopically, the tumor showed a papillary, tubular or solid pattern with occasional foam cell collections. Tumor cells were columnar or cuboidal cells with abundant eosinophilic cytoplasm (Fig. 2B-D). No psammoma bodies were found. Initial diagnosis of this case was papillary RCC type 2 and the Fuhrman nuclear grade was 3. Immunohistochemical staining showed positivity for CD10 (focal), EMA, cytokeratin 7, pan-cytokeratin, PAX-2, and vimentin and negativity for cytokeratin 20, AMACR, c-kit, TFE3, and HMB45 (Fig. 3).
RESULTS
- Recently, six RCC cases showing ALK gene rearrangement were reported by 4 independent groups.18-21 Two of the reported cases were pediatric cases (6- and 16-year-old) with a history of sickle cell trait and the RCC subtypes of these cases were renal medullary carcinoma and RCC undetermined.18,19 Renal medullary carcinoma was almost exclusively associated with sickle cell trait.2 However, the following 4 cases were adult cases without history of sickle cell trait and subtypes were papillary RCC in 3 cases and one unclassified RCC.20,21 In this study, we screened 829 RCCs and found one case with ALK immunohistochemical positivity and ALK gene rearrangement. Our case is also an adult male case without history of sickle cell trait and the initial diagnosis was papillary RCC type 2. The fusion partners of ALK in RCC were variable, with VCL in two pediatric cases and TPM3 and EML4 in two adult cases.18,19,21 In this study, we did not check the ALK fusion partner (Table 1).
- Two previous adult cases showed poor prognosis,20 and the other two adult cases had a relatively short follow-up period.21 Our patient is alive without evidence of RCC for 12 years and the follow-up period for this patient was the longest of all reported cases. This case and the previous cases suggest that RCC with ALK gene rearrangement may show variable prognosis.
- Histologic features of reported RCC cases with ALK rearrangement are variable. Two previous pediatric cases showed typical histologic features of renal medullary carcinoma19 or mixed features of renal medullary carcinoma, chromophobe RCC and transitional cell carcinoma.18 Three previous adult cases had the histologic features of papillary RCC and another case showed papillary, tubular or cribriform growth pattern. Psammoma bodies were found in all previous adult cases and foam cell collection was described in 3 out of 4 cases.20,21 Our case also showed the features of papillary RCC having foam cell collections but psammoma bodies were not found. Excluding the two pediatric cases with sickle cell trait, all four previous adult cases and our case showed the papillary growth pattern.20,21
- Two previous studies included large-scale screenings to detect RCC with ALK gene alteration. ALK rearrangement was mainly found in papillary RCC in these two studies, and they could not find ALK rearrangement in 505 clear cell RCC and 34 chromophobe RCC.20,21 We also could not find ALK expression in 689 clear cell RCCs, 73 chromophobe RCCs, and 14 rare RCC subtypes by immunohistochemistry. Taken together, the results of previous reports and this study indicate that RCC with ALK rearrangement may be a particular subset of papillary RCC.
- It is difficult to define the frequency of RCC with ALK rearrangement. A previous large-scale study showed the very low incidence of RCC with ALK rearrangement (0.56% of all RCCs), but they occupied 3.7% of non-clear cell and non-chromophobe RCC (2/32).21
- Previous adult RCC cases with ALK rearrangement showed similar immunohistochemical staining results, with positivity for cytokeratin, cytokeratin 7, and focal positive or negative for CD10.20,21 These immunohistochemical results are compatible with those of typical papillary RCCs and our case also showed similar results of positivity for cytokeratin and cytokeratin 7, and focal positivity for CD10.
- Many targeted therapies have been used for advanced RCC. ALK rearrangement in RCC may add another option for treatment of advanced RCC. ALK inhibition has been tried for treatment of other tumors with ALK rearrangement and some successful results has been reported.10,11 Therefore, we can try ALK inhibition in the treatment of advanced RCC with ALK rearrangement. FISH has been accepted as a standard method for detecting ALK gene rearrangement, but also has disadvantages in terms of cost and time. Instead of FISH, ALK immunohistochemistry can be a reliable screening tool.24 We would have found our case from screening based on ALK immunohistochemistry, which suggests that ALK immunohistochemistry is useful for the detection of RCC with ALK rearrangement.
- In conclusion, as far as we know, we have found the first RCC case with ALK gene rearrangement in Korean patients among 829 RCCs. This case showed similar histological and immunohistochemical features to previous adult cases with ALK rearrangement, and showed a relatively good prognosis.
DISCUSSION
Acknowledgments
Acknowledgments
- 1. Jung KW, Won YJ, Kong HJ, Oh CM, Seo HG, Lee JS. Cancer statistics in Korea: incidence, mortality, survival and prevalence in 2010. Cancer Res Treat 2013; 45: 1–14. PMID: 23613665. ArticlePubMedPMCPDF
- 2. Eble JN, Sauter G, Epstein JI, Sesterhenn IA. Pathology and genetics of tumours of the urinary system and male genital organs. Lyon: IARC Press, 2004; 12–39.
- 3. Patard JJ, Leray E, Rioux-Leclercq N, et al. Prognostic value of histologic subtypes in renal cell carcinoma: a multicenter experience. J Clin Oncol 2005; 23: 2763–2771. PMID: 15837991. ArticlePubMed
- 4. Singer EA, Gupta GN, Srinivasan R. Targeted therapeutic strategies for the management of renal cell carcinoma. Curr Opin Oncol 2012; 24: 284–290. PMID: 22343386. ArticlePubMedPMC
- 5. Morris SW, Kirstein MN, Valentine MB, et al. Fusion of a kinase gene, ALK, to a nucleolar protein gene, NPM, in non-Hodgkin's lymphoma. Science 1994; 263: 1281–1284. PMID: 8122112. ArticlePubMed
- 6. Laurent C, Do C, Gascoyne RD, et al. Anaplastic lymphoma kinase-positive diffuse large B-cell lymphoma: a rare clinicopathologic entity with poor prognosis. J Clin Oncol 2009; 27: 4211–4216. PMID: 19636007. ArticlePubMed
- 7. Wang WY, Gu L, Liu WP, Li GD, Liu HJ, Ma ZG. ALK-positive extramedullary plasmacytoma with expression of the CLTC-ALK fusion transcript. Pathol Res Pract 2011; 207: 587–591. PMID: 21855232. ArticlePubMed
- 8. Cook JR, Dehner LP, Collins MH, et al. Anaplastic lymphoma kinase (ALK) expression in the inflammatory myofibroblastic tumor: a comparative immunohistochemical study. Am J Surg Pathol 2001; 25: 1364–1371. PMID: 11684952. ArticlePubMed
- 9. Chen Y, Takita J, Choi YL, et al. Oncogenic mutations of ALK kinase in neuroblastoma. Nature 2008; 455: 971–974. PMID: 18923524. ArticlePubMed
- 10. Kwak EL, Bang YJ, Camidge DR, et al. Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N Engl J Med 2010; 363: 1693–1703. PMID: 20979469. ArticlePubMedPMC
- 11. Butrynski JE, D'Adamo DR, Hornick JL, et al. Crizotinib in ALK-rearranged inflammatory myofibroblastic tumor. N Engl J Med 2010; 363: 1727–1733. PMID: 20979472. ArticlePubMedPMC
- 12. Galkin AV, Melnick JS, Kim S, et al. Identification of NVP-TAE684, a potent, selective, and efficacious inhibitor of NPM-ALK. Proc Natl Acad Sci U S A 2007; 104: 270–275. PMID: 17185414. ArticlePubMed
- 13. Cerchietti L, Damm-Welk C, Vater I, et al. Inhibition of anaplastic lymphoma kinase (ALK) activity provides a therapeutic approach for CLTC-ALK-positive human diffuse large B cell lymphomas. PLoS One 2011; 6: e18436PMID: 21494621. ArticlePubMedPMC
- 14. Azarova AM, Gautam G, George RE. Emerging importance of ALK in neuroblastoma. Semin Cancer Biol 2011; 21: 267–275. PMID: 21945349. ArticlePubMedPMC
- 15. Kim H, Yoo SB, Choe JY, et al. Detection of ALK gene rearrangement in non-small cell lung cancer: a comparison of fluorescence in situ hybridization and chromogenic in situ hybridization with correlation of ALK protein expression. J Thorac Oncol 2011; 6: 1359–1366. PMID: 21587085. ArticlePubMed
- 16. Paik JH, Choe G, Kim H, et al. Screening of anaplastic lymphoma kinase rearrangement by immunohistochemistry in non-small cell lung cancer: correlation with fluorescence in situ hybridization. J Thorac Oncol 2011; 6: 466–472. PMID: 21258247. ArticlePubMed
- 17. Paik JH, Choi CM, Kim H, et al. Clinicopathologic implication of ALK rearrangement in surgically resected lung cancer: a proposal of diagnostic algorithm for ALK-rearranged adenocarcinoma. Lung Cancer 2012; 76: 403–409. PMID: 22129856. ArticlePubMed
- 18. Debelenko LV, Raimondi SC, Daw N, et al. Renal cell carcinoma with novel VCL-ALK fusion: new representative of ALK-associated tumor spectrum. Mod Pathol 2011; 24: 430–442. PMID: 21076462. ArticlePubMed
- 19. Mariño-Enríquez A, Ou WB, Weldon CB, Fletcher JA, Pérez-Atayde AR. ALK rearrangement in sickle cell trait-associated renal medullary carcinoma. Genes Chromosomes Cancer 2011; 50: 146–153. PMID: 21213368. ArticlePubMed
- 20. Sukov WR, Hodge JC, Lohse CM, et al. ALK alterations in adult renal cell carcinoma: frequency, clinicopathologic features and outcome in a large series of consecutively treated patients. Mod Pathol 2012; 25: 1516–1525. PMID: 22743654. ArticlePubMed
- 21. Sugawara E, Togashi Y, Kuroda N, et al. Identification of anaplastic lymphoma kinase fusions in renal cancer: large-scale immunohistochemical screening by the intercalated antibody-enhanced polymer method. Cancer 2012; 118: 4427–4436. PMID: 22252991. ArticlePubMed
- 22. Kim SH, Choi Y, Jeong HY, Lee K, Chae JY, Moon KC. Usefulness of a break-apart FISH assay in the diagnosis of Xp11.2 translocation renal cell carcinoma. Virchows Arch 2011; 459: 299–306. PMID: 21773754. ArticlePubMed
- 23. Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A. AJCC cancer staging manual. 7th ed. New York: Springer, 2010.
- 24. Conklin CM, Craddock KJ, Have C, Laskin J, Couture C, Ionescu DN. Immunohistochemistry is a reliable screening tool for identification of ALK rearrangement in non-small-cell lung carcinoma and is antibody dependent. J Thorac Oncol 2013; 8: 45–51. PMID: 23196275. ArticlePMC
References
Fig. 1Anaplastic lymphoma kinase (ALK) immunohistochemistry and fluorescence in situ hybridization. (A) Tumor cells show diffuse strong positivity for ALK in immunohistochemistry. (B) FISH analysis using dual color ALK break-apart probe shows one red, one green, and one fused signal indicating one rearranged ALK gene and one intact ALK gene.
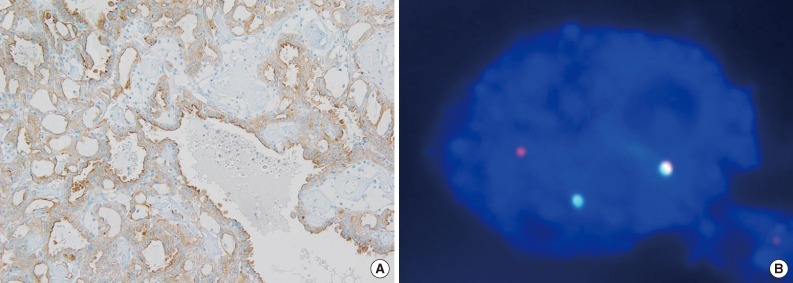

Fig. 2Pathologic features of renal cell carcinoma showing anaplastic lymphoma kinase (ALK) rearrangement. (A) Grossly, the tumor is a well-circumscribed, partially cystic mass. Microscopically, the tumor shows a papillary (B), tubular or solid pattern (C) with occasional foam cell collections (D).
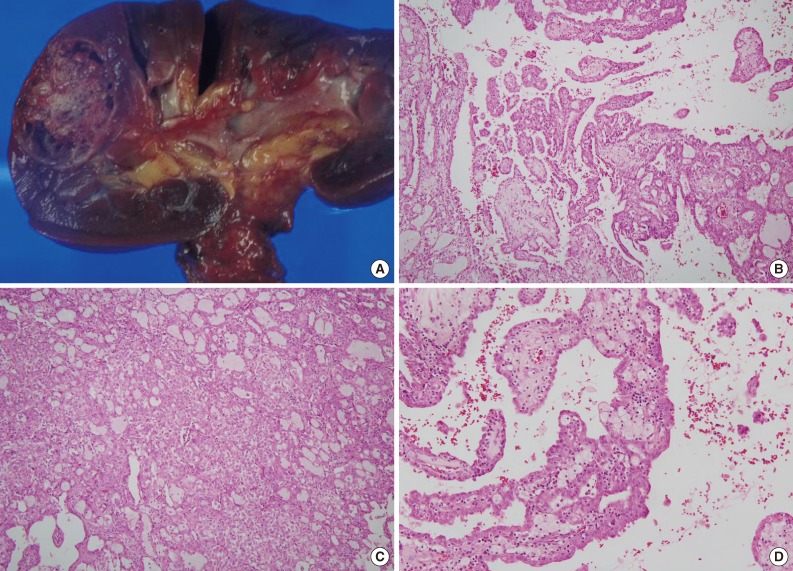

Fig. 3Immunohistochemical findings of renal cell carcinoma showing anaplastic lymphoma kinase (ALK) rearrangement. The tumor cells are positive for cytokeratin 7 (A), PAX-2 (B), and focal positive for CD10 (C) and negative for alpha-methylacyl-CoA racemase (D).
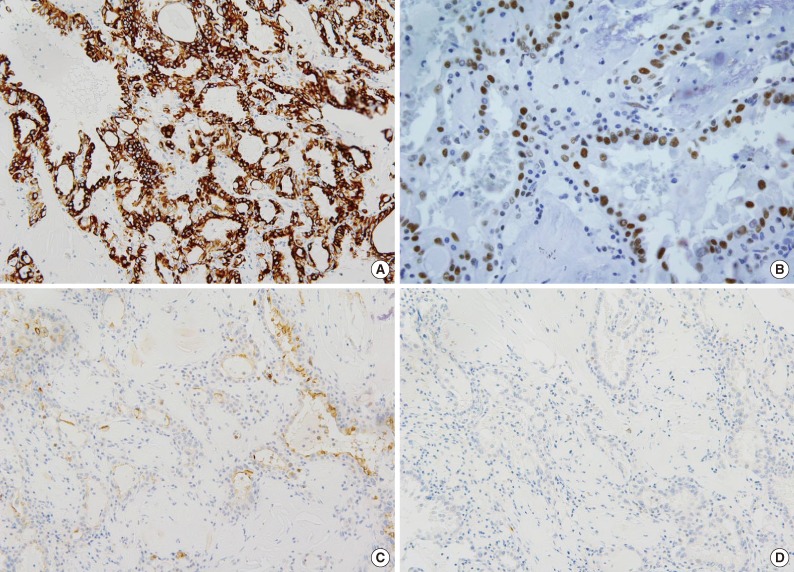

Table 1.Seven ALK-positive renal cell carcinomas
| Case No. | Age (yr) | Sickle cell trait | Subtype | Size (cm) | Fuhrman grade | TNM stage23 | Follow-up | Fusion partner | Reference |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 16/M | Yes | Indeterminate | 6.2 | Not mentioned | T1bN1M0 | 0.75 yr, alive, no evidence of disease | VCL | Debelenko et al. [18] |
| 2 | 6/M | Yes | Medullary | 4.5 | Not mentioned | T1bN0M0 | 1.75 yr, alive, no evidence of disease | VCL | Marino-Enriquez et al. [19] |
| 3 | 36/F | No | Unclassified | 4 | Not mentioned | T1aN0M0 | 2 yr, alive, no evidence of disease | TPM3 | Sugawara et al. [21] |
| 4 | 53/F | No | A: papillary, | A: 2.5, | Not mentioned | T1aN0M0 | 7 yr, alive, no evidence of disease | EML4 | Sugawara et al. [21] |
| B: clear cell (double cancer) | B: 0.6 | ||||||||
| 5 | 61/M | No | Papillary | 5 | 2 | T1bNXM0 | 4 yr, death from disease | Unchecked | Sukov et al. [20] |
| 6 | 59/M | No | Papillary | 5.5 | 3 | T1bNXM0 | 1.4 yr, death from disease | Unchecked | Sukov et al. [20] |
| 7 | 44/M | No | Papillary | 3 | 3 | T1aN0M0 | 12 yr, alive, no evidence of disease | Unchecked | Present case |
Figure & Data
References
Citations
Citations to this article as recorded by 

- Activity of ALK Inhibitors in Renal Cancer with ALK Alterations: A Systematic Review
Giovanni Maria Iannantuono, Silvia Riondino, Stefano Sganga, Mario Roselli, Francesco Torino
International Journal of Molecular Sciences.2022; 23(7): 3995. CrossRef - Novel, emerging and provisional renal entities: The Genitourinary Pathology Society (GUPS) update on renal neoplasia
Kiril Trpkov, Sean R. Williamson, Anthony J. Gill, Adebowale J. Adeniran, Abbas Agaimy, Reza Alaghehbandan, Mahul B. Amin, Pedram Argani, Ying-Bei Chen, Liang Cheng, Jonathan I. Epstein, John C. Cheville, Eva Comperat, Isabela Werneck da Cunha, Jennifer B
Modern Pathology.2021; 34(6): 1167. CrossRef - ESC, ALK, HOT and LOT: Three Letter Acronyms of Emerging Renal Entities Knocking on the Door of the WHO Classification
Farshid Siadat, Kiril Trpkov
Cancers.2020; 12(1): 168. CrossRef - ALK-rearranged renal cell carcinoma with a novel PLEKHA7-ALK translocation and metanephric adenoma-like morphology
Jen-Fan Hang, Hsiao-Jen Chung, Chin-Chen Pan
Virchows Archiv.2020; 476(6): 921. CrossRef - Characteristics of Renal Cell Carcinoma Harboring TPM3-ALK Fusion
Chang Gok Woo, Seok Jung Yun, Seung-Myoung Son, Young Hyun Lim, Ok-Jun Lee
Yonsei Medical Journal.2020; 61(3): 262. CrossRef - Report From the International Society of Urological Pathology (ISUP) Consultation Conference on Molecular Pathology of Urogenital Cancers
Sean R. Williamson, Anthony J. Gill, Pedram Argani, Ying-Bei Chen, Lars Egevad, Glen Kristiansen, David J. Grignon, Ondrej Hes
American Journal of Surgical Pathology.2020; 44(7): e47. CrossRef - ALK rearranged renal cell carcinoma (ALK-RCC): a multi-institutional study of twelve cases with identification of novel partner genes CLIP1, KIF5B and KIAA1217
Naoto Kuroda, Kiril Trpkov, Yuan Gao, Maria Tretiakova, Yajuan J. Liu, Monika Ulamec, Kengo Takeuchi, Abbas Agaimy, Christopher Przybycin, Cristina Magi-Galluzzi, Soichiro Fushimi, Fumiyoshi Kojima, Malthide Sibony, Jen-Fan Hang, Chin-Chen Pan, Asli Yilma
Modern Pathology.2020; 33(12): 2564. CrossRef - ALK rearrangement in TFE3-positive renal cell carcinoma: Alternative diagnostic option to exclude Xp11.2 translocation carcinoma
Yiqi Zhu, Ning Liu, Wei Guo, Xiaohong Pu, Hongqian Guo, Weidong Gan, Dongmei Li
Pathology - Research and Practice.2020; 216(12): 153286. CrossRef - New and emerging renal entities: a perspective post‐WHO 2016 classification
Kiril Trpkov, Ondřej Hes
Histopathology.2019; 74(1): 31. CrossRef - Lack of expression of ALK and CD30 in breast carcinoma by immunohistochemistry irrespective of tumor characteristics
Samer Nassif, Ziad M. El-Zaatari, Michel Attieh, Maya Hijazi, Najla Fakhreddin, Tarek Aridi, Fouad Boulos
Medicine.2019; 98(32): e16702. CrossRef - Targeted next-generation sequencing revealed distinct clinicopathologic and molecular features of VCL-ALK RCC: A unique case from an older patient without clinical evidence of sickle cell trait
Xiao-tong Wang, Ru Fang, Sheng-bing Ye, Ru-song Zhang, Rui Li, Xuan Wang, Rong-hao Ji, Zhen-feng Lu, Heng-hui Ma, Xiao-jun Zhou, Qiu-yuan Xia, Qiu Rao
Pathology - Research and Practice.2019; 215(11): 152651. CrossRef - ALK-rearranged renal cell carcinomas in Polish population
Adam Gorczynski, Piotr Czapiewski, Aleksandra Korwat, Lukasz Budynko, Monika Prelowska, Krzysztof Okon, Wojciech Biernat
Pathology - Research and Practice.2019; 215(12): 152669. CrossRef - ALK-TPM3 rearrangement in adult renal cell carcinoma: a case report and literature review
Jing Yang, Lei Dong, Hong Du, Xiu-bo Li, Yan-xiao Liang, Guo-rong Liu
Diagnostic Pathology.2019;[Epub] CrossRef - Molecular Genetics of Renal Cell Tumors: A Practical Diagnostic Approach
Reza Alaghehbandan, Delia Perez Montiel, Ana Silvia Luis, Ondrej Hes
Cancers.2019; 12(1): 85. CrossRef - ALK-TPM3 rearrangement in adult renal cell carcinoma: Report of a new case showing loss of chromosome 3 and literature review
Yohan Bodokh, Damien Ambrosetti, Valérie Kubiniek, Branwel Tibi, Matthieu Durand, Jean Amiel, Morgane Pertuit, Anne Barlier, Florence Pedeutour
Cancer Genetics.2018; 221: 31. CrossRef - Prognostic implications of polycomb proteins ezh2, suz12, and eed1 and histone modification by H3K27me3 in sarcoma
Yong Jin Cho, Soo Hee Kim, Eun Kyung Kim, Jung Woo Han, Kyoo-Ho Shin, Hyuk Hu, Kyung Sik Kim, Young Deuk Choi, Sunghoon Kim, Young Han Lee, Jin-Suck Suh, Joong Bae Ahn, Hyun Cheol Chung, Sung Hoon Noh, Sun Young Rha, Sung-Taek Jung, Hyo Song Kim
BMC Cancer.2018;[Epub] CrossRef - Responses to Alectinib in ALK-rearranged Papillary Renal Cell Carcinoma
Sumanta K. Pal, Paulo Bergerot, Nazli Dizman, Cristiane Bergerot, Jacob Adashek, Russell Madison, Jon H. Chung, Siraj M. Ali, Jeremy O. Jones, Ravi Salgia
European Urology.2018; 74(1): 124. CrossRef - Genetic analysis and clinicopathological features of ALK‐rearranged renal cell carcinoma in a large series of resected Chinese renal cell carcinoma patients and literature review
Wenjuan Yu, Yuewei Wang, Yanxia Jiang, Wei Zhang, Yujun Li
Histopathology.2017; 71(1): 53. CrossRef - A case of anaplastic lymphoma kinase‐positive renal cell carcinoma coincident with Hodgkin lymphoma
Yuzo Oyama, Haruto Nishida, Takahiro Kusaba, Hiroko Kadowaki, Motoki Arakane, Tsutomu Daa, Dai Watanabe, Yasuyuki Akita, Fuminori Sato, Hiromitsu Mimata, Shigeo Yokoyama
Pathology International.2017; 67(12): 626. CrossRef - Clinicopathologic and Molecular Pathology of Collecting Duct Carcinoma and Related Renal Cell Carcinomas
An Na Seo, Ghilsuk Yoon, Jae Y. Ro
Advances in Anatomic Pathology.2017; 24(2): 65. CrossRef - The role of the polycomb repressive complex pathway in T and NK cell lymphoma: biological and prognostic implications
Soo Hee Kim, Woo Ick Yang, Yoo Hong Min, Young Hyeh Ko, Sun Och Yoon
Tumor Biology.2016; 37(2): 2037. CrossRef - New and emerging renal tumour entities
Naoto Kuroda, Ondřej Hess, Ming Zhou
Diagnostic Histopathology.2016; 22(2): 47. CrossRef - ALK‐rearranged renal cell carcinomas in children
Mariana M. Cajaiba, Lawrence J. Jennings, Stephen M. Rohan, Antonio R. Perez‐Atayde, Adrian Marino‐Enriquez, Jonathan A. Fletcher, James I. Geller, Katrin M. C. Leuer, Julia A. Bridge, Elizabeth J. Perlman
Genes, Chromosomes and Cancer.2016; 55(5): 442. CrossRef - Two Cases of Renal Cell Carcinoma Harboring a Novel STRN-ALK Fusion Gene
Hironori Kusano, Yuki Togashi, Jun Akiba, Fukuko Moriya, Katsuyoshi Baba, Naomi Matsuzaki, Yoshiaki Yuba, Yusuke Shiraishi, Hiroshi Kanamaru, Naoto Kuroda, Seiji Sakata, Kengo Takeuchi, Hirohisa Yano
American Journal of Surgical Pathology.2016; 40(6): 761. CrossRef - Expanding the spectrum of ALK‐rearranged renal cell carcinomas in children: Identification of a novel HOOK1‐ALK fusion transcript
Mariana M. Cajaiba, Lawrence J. Jennings, David George, Elizabeth J. Perlman
Genes, Chromosomes and Cancer.2016; 55(10): 814. CrossRef - TFE3-positive renal cell carcinomas are not always Xp11 translocation carcinomas: Report of a case with a TPM3-ALK translocation
Paul Scott Thorner, Mary Shago, Paula Marrano, Furqan Shaikh, Gino R. Somers
Pathology - Research and Practice.2016; 212(10): 937. CrossRef - ALK rearrangements-associated renal cell carcinoma (RCC) with unique pathological features in an adult
Marie Jeanneau, Valerie Gregoire, Claude Desplechain, Fabienne Escande, Dan Petre Tica, Sebastien Aubert, Xavier Leroy
Pathology - Research and Practice.2016; 212(11): 1064. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure

 E-submission
E-submission