Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 51(2); 2017 > Article
-
Original Article
Higher Expression of Toll-like Receptors 3, 7, 8, and 9 in Pityriasis Rosea - Mostafa Abou El-Ela, Mohamed El-Komy, Rania Abdel Hay,, Rehab Hegazy, Amin Sharobim1, Laila Rashed2, Khalda Amr3
-
Journal of Pathology and Translational Medicine 2017;51(2):148-151.
DOI: https://doi.org/10.4132/jptm.2016.09.09
Published online: February 13, 2017
Department of Dermatology, Faculty of Medicine, Cairo University, Cairo, Egypt
1Department of Dermatology, National Research Center (NRC), Cairo University, Cairo, Egypt
2Department of Clinical Biochemistry, Faculty of Medicine, Cairo University, Cairo, Egypt
3Department of Molecular Genetics, National Research Center (NRC), Cairo University, Cairo, Egypt
- Corresponding Author Rania Abdel Hay, MD Department of Dermatology, Faculty of Medicine, Cairo University, 13th Abrag Othman, Kournish el Maadi, Cairo 11431, Egypt Tel: +20-1006084132 Fax: +20-23596724 E-mail: 'raniamounir@kasralainy.edu.eg'
© 2017 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Pityriasis rosea (PR) is a common papulosquamous skin disease in which an infective agent may be implicated. Toll-like receptors (TLRs) play an important role in immune responses and in the pathophysiology of inflammatory skin diseases. Our aim was to determine the possible roles of TLRs 3, 7, 8, and 9 in the pathogenesis of PR.
-
Methods
- Twenty-four PR patients and 24 healthy individuals (as controls) were included in this case control study. All recruits were subjected to routine laboratory investigations. Biopsies were obtained from one active PR lesion and from healthy skin of controls for the detection of TLR 3, 7, 8, and 9 gene expression using real-time polymerase chain reaction.
-
Results
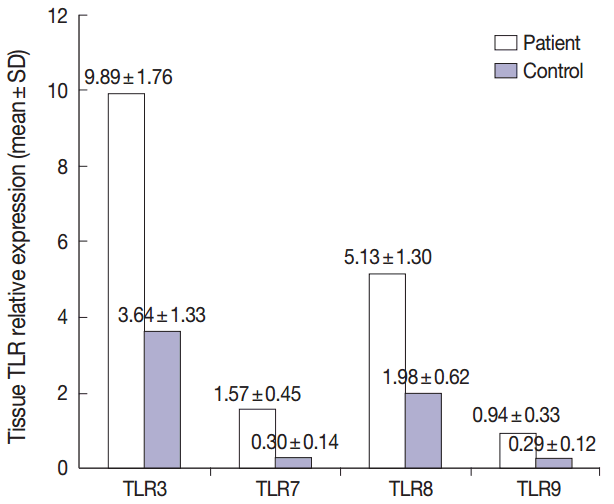
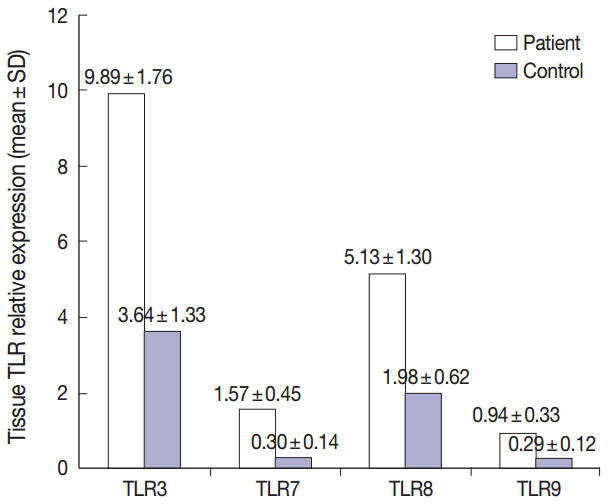
- This study included 24 patients (8 females and 16 males) with active PR lesions, with a mean age of 28.62 years. Twenty four healthy age- and sex-matched individuals were included as controls (8 females and 16 males, with a mean age of 30.83 years). The results of the routine laboratory tests revealed no significant differences between both groups. Significantly elevated expression of all studied TLRs were detected in PR patients relative to healthy controls (p < .001).
-
Conclusions
- TLRs 3, 7, 8, and 9 might be involved in the pathogenesis of PR.
- The current case control study was approved by the Research Ethical Committee Office (REC), Department of Dermatology, Faculty of Medicine, Cairo University. All participants provided full informed written consent prior to this study.
- This study included 24 patients with classic active PR and 24 age- and sex-matched and apparently healthy individuals serving as controls. All participants were recruited between May 2012 and January 2013 from the dermatology outpatient clinic of Kasr Al Ainy Hospital.
- All patients in the PR group had classical findings of PR and were in the active stage of the disease (having active lesions appearing within the same week of their visit to the clinic). All participants were otherwise healthy and immunocompetent, none had a recent history of immunization, and none were on systemic steroids or other immunosuppressive therapy.
- All recruits were subjected to routine laboratory investigations including hemoglobin (Hb), white blood cells count, erythrocyte sedimentation rate first hour (ESR1), erythrocyte sedimentation rate second hour (ESR2), alanine aminotransferase (ALT), aspartate aminotransferase (AST), and urea and creatinine levels.
- A 4-mm punch biopsy was obtained from the active edge of one of the lesions from each patient and from the apparently healthy skin of the trunk from each control. The biopsies were stored at –70˚C until used for measurement of TLR mRNA.
- Detection of TLR 3, 7, 8, and 9 gene expression levels using real-time polymerase chain reaction
- Total RNA was extracted from skin tissue (TRIzol reagent, Invitrogen, Karlsruhe, Germany), and the quantity and purity of the extracted RNA were assessed by measuring absorbance at 260 nm and the ratio of absorbance at 260 nm to absorbance at 280 nm (A260/A280), using an ultraviolet spectrophotometer (NanoDrop Inc., Wilmington, DE, USA). Only samples with an A260/A280 ratio under 1.8 were considered valid for real-time polymerase chain reaction (RT-PCR). Reverse transcription of 1 μg of RNA into cDNA was performed (First Strand cDNA Synthesis Kit, TaKaRa, Bio Inc., Shiga, Japan) in accordance with the manufacturer’s instructions.
- Expression of the target gene TLR was quantified using the comparative threshold cycle (Ct) method, with the amount of target mRNA normalized to an internal control (glyceraldehyde phosphate dehydogenase [GAPDH]). RT-PCR was performed using a commercial kit (Light Cycler Fast Start DNA SYBR-Green I Kit, Roche Applied Sciences, Mannheim, Germany) in accordance with the provided protocol. Briefly, 10 μL amplification mixtures were prepared, containing the equivalent of 8 ng of reverse-transcribed RNA, along with 300 M primers (sequences of the PCR primer pairs used for each gene are shown in Table 1). Reactions were run on a cetection system (ABI Prism 7900 HT, Applied Biosystems, Foster City, CA, USA). PCR parameters were one cycle at 95˚C for 10 minutes, followed by 40 cycles at 94˚C for 15 seconds and 60˚C for 1 minute. Data were analyzed and quantified using ABI Prism sequence detection system software (Sequence Detection Software v1.7, PE Applied Biosystems). Relative expression of studied genes was calculated using the comparative threshold cycle method, and all values were normalized to GAPDH genes [9]. The level of expression of each target gene was normalized relative to the expression of GAPDH mRNA in that sample using the ΔCt method. Relative differences in gene expression between groups were determined using the comparative Ct (ΔΔCt) method, and fold expression was calculated as 2–ΔΔCt, where ΔΔCt represents ΔCt values normalized relative to the mean ΔCt of control samples.
- Statistical methods
- Data were described in terms of mean±standard deviation (±SD), frequencies (number of cases) and relative frequencies (%) when appropriate. Comparisons between groups were done using T-tests for all quantitative variables, and chi-square (χ2) tests for categorical data (sex distribution). Correlations between various variables were calculated using Pearson moment correlation equations for linear relationships. p<.05 was considered statistically significant. All statistical calculations were done using the computer program Microsoft Excel (Microsoft Corporation, New York, NY, USA) and SPSS ver. 19 for Microsoft Windows (SPSS Inc., Chicago, IL, USA).
MATERIALS AND METHODS
- This case control study included 24 patients (8 females [33.3%] and 16 males [66.7%]) with classic active PR developing within the 2 weeks prior to recruitment. Their age ranged from 19 to 45 years with mean±SD of 28.62±8.30. Twenty-four apparently healthy age- and sex-matched individuals (p=.370 and p>.990, respectively) were also included in this study as controls. The controls included eight females (33.3%) and 16 males (66.7%), and their age ranged from 19–46 years with mean±SD of 30.83±8.59.
- Routine laboratory data
- Comparing routine laboratory findings between the patient and control groups revealed no significant differences (using T-test) (Table 2). In the patient group, 14 patients had anemia (low Hb), none of the patients had leukocytosis or leucopenia, 18 patients showed elevated ESR1 and ESR2, none of the patients had elevated ALT or AST, and none showed elevated urea or creatinine levels. In the control group, 14 controls had anemia (low Hb), two controls showed leucopenia and none had leukocytosis, 15 controls showed elevated ESR1 and ESR2, none of the controls had elevated ALT or AST, and none showed elevated urea or creatinine levels.
- Histopathologic findings among PR patients included focal parakeratosis, absent granular cell layer, mild acanthosis, mild spongiosis, papillary dermal edema, perivascular and superficial dermal interstitial infiltrate of lymphocytes and histiocytes, and focal extravasation of erythrocytes as previously described [10], confirming the diagnosis of PR.
- TLRs expression
- Summary of the descriptive data of different studied TLRs in both groups were presented in Fig. 1. Comparing the TLR 3, 7, 8, and 9 expression levels between the patient and control groups revealed that the mean levels of all studied TLRs were significantly higher in patients relative to healthy controls (p<.001 for all, using T-test) (Fig. 1).
- Our results revealed nonsignificant correlations between the studied TLRs in the patient group (using Pearson correlation test).
RESULTS
- The current study highlights the possible roles played by different TLR pathways in the pathogenesis of PR. In this study, a significantly elevated expression of the studied TLRs 3, 7, 8, and 9 was documented in PR cases relative to healthy controls.
- The results of the current study are in accordance with another [8], which found the expression of TLRs 3, 7, and 9 to be significantly elevated in the peripheral blood (PB) lymphocytes of patients with PR relative to normal controls. However, unlike in our study, that study did not find elevated levels in the skin. This discrepancy between skin and PB lymphocytes may support the concept of PR being induced through a systemic insult to the body, most likely an infective agent, with the primary and early burden on the immune system.
- Our results revealed non-significant correlations among the studied TLRs in the patient group. This could suggest that each TLR can play an individual role in the pathogenesis of PR.
- TLRs 3, 7, 8, and 9 have been shown to be involved in responses to viral infection [11], but no studies have referred to their involvement with HHV. The use of TLRs 3, 7, 8, and 9 analogs as antiviral therapy supports the role of TLR in viral infections [12,13].
- The possible role of TLRs in PR can be attributed to several mechanisms. Vercammen et al. [14] demonstrated that TLR 3 triggering activates specific signaling pathways that mount an effective immune response through the induction of cytokines and other pro-inflammatory mediators. These mediators may participate in enhancing inflammation in the affected area of the PR. Renn et al. [15] also reported that after the stimulation of TLR 3, and to a lesser extent TLRs 7 and 8, langerhans cells are stimulated, which were found to be increased in lesions of PR; langerhans cells produce large amounts of chemoattractants, which may participate in the inflammation present in PR.
- However, the fact that TLRs are involved with the initiation of innate and acquired immune responses for many other pathogens precludes us from asserting that their high expression in PR points to an infective etiology.
- In conclusion, the current study indicated that TLRs 3, 7, 8 and 9 have a positive role in PR. Studying the link between HHV 6 and 7 and TLRs in cases of PR is part of our ongoing research in order to report a possible link between the TLRs and HHV in the pathogenesis of PR and to detect possible triggering factors for HHV reinfection or reactivation in PR.
DISCUSSION
-
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Notes
Values are presented as mean ± standard deviation (range).
Normal routine laboratory reference values: hemoglobin (Hb; male: 13–18 mg/female: 12–17 mg), white blood cells (WBCs; 4,000–12,000 mm3), erythrocyte sedimentation rate first hour (ESR1; up to 8 mm/hr), erythrocyte sedimentation rate second hour (ESR2; up to 18 mm/hr), alanine aminotransferase (ALT; 5–40 U/L), aspartate aminotransferase (AST; 7–56 U/L), urea (20–50 mg/dL), creatinine (0.5–1.5 mg/dL).
- 1. Chuh A, Chan H, Zawar V. Pityriasis rosea: evidence for and against an infectious aetiology. Epidemiol Infect 2004; 132: 381–90. ArticlePubMedPMC
- 2. Drago F, Vecchio F, Rebora A. Use of high-dose acyclovir in pityriasis rosea. J Am Acad Dermatol 2006; 54: 82–5. ArticlePubMed
- 3. Watanabe T, Kawamura T, Jacob SE, et al. Pityriasis rosea is associated with systemic active infection with both human herpesvirus-7 and human herpesvirus-6. J Invest Dermatol 2002; 119: 793–7. ArticlePubMed
- 4. Drago F, Ciccarese G, Broccolo F, Cozzani E, Parodi A. Pityriasis rosea in children: clinical features and laboratory investigations. Dermatology 2015; 231: 9–14. ArticlePubMed
- 5. Broccolo F, Drago F, Careddu AM, et al. Additional evidence that pityriasis rosea is associated with reactivation of human herpesvirus-6 and -7. J Invest Dermatol 2005; 124: 1234–40. ArticlePubMed
- 6. Trinchieri G, Sher A. Cooperation of Toll-like receptor signals in innate immune defence. Nat Rev Immunol 2007; 7: 179–90. ArticlePubMed
- 7. Wagner H. The immunobiology of the TLR9 subfamily. Trends Immunol 2004; 25: 381–6. ArticlePubMed
- 8. Yang F, Han XP. Expression of Toll-like receptor 3, 7 and 9 in peripheral blood lymphocytes from patients with pityriasis rosea. Chin J Dermatol 2012; 45: 130–2.
- 9. Pfaffl MW. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 2001; 29: e45.ArticlePubMedPMC
- 10. Ataseven A, Kurtipek GS, Akyurek FT, Kucukosmanoglu I, Dilek N. Unilateral pityriasis rosea. Indian Dermatol Online J 2014; 5: 528–9. ArticlePubMedPMC
- 11. Carty M, Bowie AG. Recent insights into the role of Toll-like receptors in viral infection. Clin Exp Immunol 2010; 161: 397–406. ArticlePubMedPMC
- 12. Miller RL, Meng TC, Tomai MA. The antiviral activity of Toll-like receptor 7 and 7/8 agonists. Drug News Perspect 2008; 21: 69–87. ArticlePubMed
- 13. Lau YF, Tang LH, Ooi EE. A TLR3 ligand that exhibits potent inhibition of influenza virus replication and has strong adjuvant activity has the potential for dual applications in an influenza pandemic. Vaccine 2009; 27: 1354–64. ArticlePubMedPMC
- 14. Vercammen E, Staal J, Beyaert R. Sensing of viral infection and activation of innate immunity by toll-like receptor 3. Clin Microbiol Rev 2008; 21: 13–25. ArticlePubMedPMC
- 15. Renn CN, Sanchez DJ, Ochoa MT, et al. TLR activation of Langerhans cell-like dendritic cells triggers an antiviral immune response. J Immunol 2006; 177: 298–305. ArticlePubMed
References
Figure & Data
References
Citations

- Toll-like Receptor-Mediated Immunomodulation of Th1-Type Response Stimulated by Recombinant Antigen of Type 2 Porcine Reproductive and Respiratory Syndrome Virus (PRRSV-2)
Rika Wahyuningtyas, Mei-Li Wu, Wen-Bin Chung, Hso-Chi Chaung, Ko-Tung Chang
Viruses.2023; 15(3): 775. CrossRef - Pityriasis Rosea: An Updated Review
Alexander K.C. Leung, Joseph M. Lam, Kin Fon Leong, Kam Lun Hon
Current Pediatric Reviews.2021; 17(3): 201. CrossRef - Double-blind randomized placebo-controlled trial to evaluate the efficacy and safety of short-course low-dose oral prednisolone in pityriasis rosea
Sidharth Sonthalia, Akshy Kumar, Vijay Zawar, Adity Priya, Pravesh Yadav, Sakshi Srivastava, Atula Gupta
Journal of Dermatological Treatment.2018; 29(6): 617. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure

 E-submission
E-submission