Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 48(2); 2014 > Article
-
Original Article
Characteristics of Cutaneous Lymphomas in Korea According to the New WHO-EORTC Classification: Report of a Nationwide Study - Jae Ho Han, Young-Hyeh Ko1, Yun Kyung Kang2, Wan-Seop Kim3, Yoon Jung Kim4, Insun Kim5, Hyun-Jung Kim6, Soo Kee Min7, Chan-Kum Park8, Chan-Sik Park9, Bong-Kyung Shin10, Woo Ick Yang11, Young-Ha Oh12, Jong Sil Lee13, Juhie Lee14, Tae Hui Lee15, Hyekyung Lee16, Ho Jung Lee17, Yoon Kyung Jeon18, Hee Jeong Cha19, Yoo-Duk Choi20, Chul Woo Kim18, Hematopathology Study Group of the Korean Society of Pathologists
-
Korean Journal of Pathology 2014;48(2):126-132.
DOI: https://doi.org/10.4132/KoreanJPathol.2014.48.2.126
Published online: April 28, 2014
Department of Pathology, Ajou University School of Medicine, Suwon, Korea.
1Department of Pathology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
2Department of Pathology, Inje University Seoul Paik Hospital, Seoul, Korea.
3Department of Pathology, Konkuk University School of Medicine, Seoul, Korea.
4Department of Pathology, Veterans Health Service Medical Center, Seoul, Korea.
5Department of Pathology, Korea University College of Medicine, Seoul, Korea.
6Department of Pathology, Inje University Sanggye Paik Hospital, Seoul, Korea.
7Department of Pathology, Hallym University Sacred Heart Hospital, Anyang, Korea.
8Department of Pathology, Hanyang University College of Medicine, Seoul, Korea.
9Department of Pathology, Asan Medical Center, Seoul, Korea.
10Department of Pathology, Korea University Guro Hospital, Seoul, Korea.
11Department of Pathology, Yonsei University College of Medicine, Seoul, Korea.
12Department of Pathology, Hanyang University Guri Hospital, Guri, Korea.
13Department of Pathology, Gyeongsang National University School of Medicine, Jinju, Korea.
14Department of Pathology, Kyung Hee University School of Medicine, Seoul, Korea.
15Department of Pathology, Dankook University College of Medicine, Cheonan, Korea.
16Department of Pathology, Eulji University Hospital, Daejeon, Korea.
17Department of Pathology, Eulji General Hospital, Seoul, Korea.
18Department of Pathology, Seoul National University, Seoul, Korea.
19Department of Pathology, Ulsan University College of Medicine, Ulsan, Korea.
20Department of Pathology, Chonnam National University Medical School, Gwangju, Korea.
- Corresponding Author: Chul Woo Kim, M.D. Department of Pathology, Seoul National University, 101 Daehak-ro, Jongno-gu, Seoul 110-744, Korea. Tel: +82-2-740-8267, Fax: +82-2-743-5530, cwkim@snu.ac.kr
© 2014 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Previously, cutaneous lymphomas were classified according to either the European Organization for the Research and Treatment of Cancer (EORTC) or the World Health Organization (WHO) classification paradigms. The aim of this study was to determine the relative frequency of Korean cutaneous lymphoma according to the new WHO-EORTC classification system.
-
Methods
- A total of 517 patients were recruited during a recent 5 year-period (2006-2010) from 21 institutes and classified according to the WHO-EORTC criteria.
-
Results
- The patients included 298 males and 219 females, and the mean age at diagnosis was 49 years. The lesions preferentially affected the trunk area (40.2%). The most frequent subtypes in order of decreasing prevalence were mycosis fungoides (22.2%), peripheral T-cell lymphoma (17.2%), CD30+ T-cell lymphoproliferative disorder (13.7%), and extranodal natural killer/T (NK/T) cell lymphoma, nasal type (12.0%). Diffuse large B-cell lymphoma accounted for 11.2% of cases, half of which were secondary cutaneous involvement; other types of B-cell lymphoma accounted for less than 1% of cases.
-
Conclusions
- In comparison with data from Western countries, this study revealed relatively lower rates of mycosis fungoides and B-cell lymphoma in Korean patients, as well as higher rates of subcutaneous panniculitis-like T-cell lymphoma and NK/T cell lymphoma.
- A total of 517 cases of cutaneous lymphoma diagnosed at 21 institutes in Korea between January 2006 and December 2010 were enrolled in our study. The cases were reviewed by hematopathologists and re-classified according to the new WHO-EORTC classification.2,3,4 Clinical data including age, sex, site of involvement, and primary or secondary disease were obtained. The histologic and clinical results were compared with those of previous Western studies.
MATERIALS AND METHODS
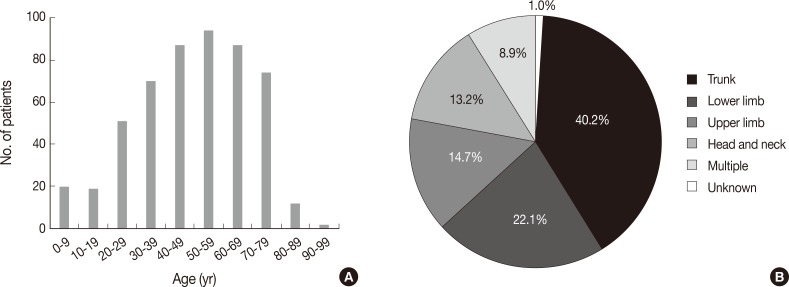
- Patients were aged between 1 and 91 years (mean, 49 years). Although any age group may be involved, cutaneous lymphoma was most common in patients in their fifth, sixth, and seventh decades, with a peak incidence at approximately 60 years of age (Fig. 1A). Patients included 298 males (57.6%) and 219 females (42.4%) (male:female ratio, 1.4:1). Lesions could be present at any site, but the trunk was the most common anatomic site (40.2%) (Fig. 1B).
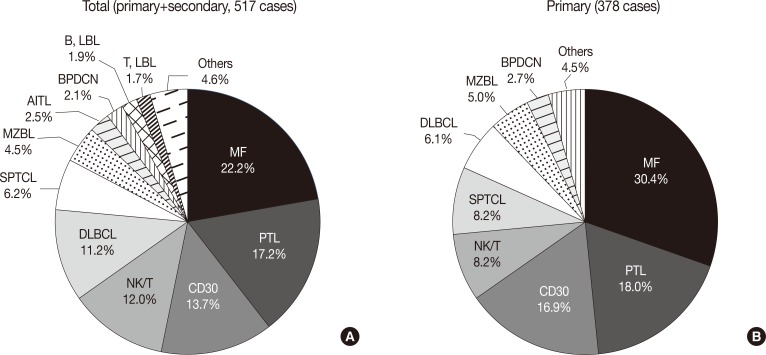
- The overall incidence of cutaneous lymphoma according to the WHO-EORTC classification is summarized in Table 1. Among a total of 517 cases of cutaneous lymphoma, primary and secondary cutaneous lymphomas accounted for 73.1% and 26.1%, respectively, while only 0.8% of cases were undetermined. Among all 517 patients, the relative frequencies of mature T-cell lymphoma, mature B-cell lymphoma, immature hematopoeitic malignancy and Hodgkin's lymphoma were 74.3%, 18.0%, 5.8%, and 0.2%, respectively. Further, for the 378 cases of primary cutaneous lymphomas, the proportion of T-cell lymphoma (82.3%) increased when compared with the proportion of total cases.
- The most frequent subtypes, in decreasing order of prevalence, were mycosis fungoides (MF) including its variants (22.2%), peripheral T-cell lymphoma (PTCL) including subtype (17.2%), CD30+ T-cell lymphoproliferative disorder (CD30+ LPD) (13.7%), and extranodal NK/T cell lymphoma, nasal type (12.0%), respectively (Fig. 2A). MF variants including pagetoid reticulosis or folliculotropic were rarely described. Of the CD30+ LPD, there were 32 (6.2%) cases of lymphomatoid papulosis (LyP) and 36 (7.0%) cases of anaplastic large cell lymphoma (ALCL). Three cases could not be subclassified, and ALCL included five systemic cases. Half of the NK/T cell lymphoma cases were secondary lymphoma. Other cutaneous T-cell lymphoma included subcutaneous panniculitis-like T-cell lymphoma (SPTCL) (6.2%), angioimmunoblastic T-cell lymphoma (AITL) (2.5%), and T lymphoblastic lymphoma (1.7%). Adult T-cell leukemia/lymphoma was rarely observed (0.2%). With respect to primary cutaneous lymphoma, the proportion of MF, CD30+ LPD and SPTCL was increased (Fig. 2B). Among B-cell lymphomas, the DLBCL was the most common lymphoma and accounted for 11.2% of cases, although half were secondary cutaneous involvement. Cutaneous marginal zone B-cell lymphoma was the second most common B-cell lymphoma (4.5%), and other types of B-cell lymphoma accounted for less than 1% of cases. Immature hematopoietic malignancies included BPDC neoplasm (11 cases, 2.1%), T-lymphoblastic leukemia/lymphoma (9 cases, 1.7%) and B-lymphoblastic leukemia/lymphoma (10 cases, 1.9%).
- The DLBCL and PTCL subtypes were rarely observed. Of the 68 primary cutaneous peripheral T-cell lymphomas, 4 cases were primary cutaneous aggressive epidermotropic CD8-positive cytotoxic T-cell lymphoma, 8 cases were gamma/delta-positive T-cell lymphoma, and 5 cases were small/medium CD4-positive T-cell lymphoma. Of the 23 cases of primary cutaneous DLBCL, 6 were categorized as DLBCL, leg type.
- Comparison of lymphoma subtype with sex and age revealed that BPDC neoplasm and T lymphoblastic lymphoma was predominantly associated with males, while CD30+ LPD, SPTCL, and lymphoblastic lymphoma occurred more often in younger individuals (Table 2). Likewise, comparison of lymphoma subtype with the involved anatomical site revealed that marginal zone B-cell lymphoma and B lymphoblastic lymphoma occurred more frequently in the head and neck (Table 3). In addition, the relative incidences of CD30+ LPD, NK/T cell lymphoma, SPTCL, and immature hematopoietic malignancies were higher while those of MF and B-cell lymphoma was lower when compared with one Western study (Table 4).5
RESULTS
- In the present study we analyzed the incidence rate of Korean cutaneous lymphoma according to the new WHO-EORTC classification. To our knowledge, only a few nation-wide studies of Korean cutaneous lymphoma exist, all of which have several limitations. Specifically, one report focused on systemic lymphoma,1 reporting an incidence of lymphoma subtype very different from the results observed in this study, such as SPTCL being the most common cutaneous lymphoma. One possible explanation for this difference is that the previous report was based on the 2001 WHO classification and described a very limited set of lymphoma subtypes. Other reports have similar limitations.7
- With respect to lymphoma subtype and sex, the results of our study were consistent with previous reports.5,8,9 Specifically, we observed a male predominance for the majority of lymphoma subtypes, especially BPDC neoplasm and T lymphoblastic lymphoma, and a female predominance for SPTCL. We also found that most patients were elderly, with a peak incidence around 60 years. As with other systemic lymphomas, aging and age-related effects such as chronic inflammation and diminished immune surveillance may contribute to lymphomagenesis.10
- Our study included primary and secondary cutaneous lymphomas. However, the distinction between primary and secondary can be difficult because of incomplete clinical evaluation. Although undetermined cases accounted for only 0.8% of the study population, more problematic cases involving distinction between primary and secondary cutaneous lymphoma may exist. Secondary cutaneous involvement was relatively frequent in NK/T cell lymphoma, AITL, DLBCL, and lymphoblastic lymphoma. Furthermore, almost all of the patients in our study with AITL had systemic lymphoma, indicating secondary cutaneous involvement. AITL skin lesions are nonspecific subtle perivascular lymphoid cell infiltrates without atypia. Further, it is not clear whether such cutaneous manifestations are due to the tumor cell itself or secondary cytokine effects. Thus, immunohistochemical staining for follicular helper T cell markers such as PD-1 and CXCL13 may be helpful in making decisions.
- In our study, T cell lymphoma comprised the majority (74.3%) of cutaneous lymphomas, similar to studies conducted in Western countries.3,8,11 MF was the most common primary cutaneous lymphoma. MF displays various histologic appearances mimicking ALCL and PTCL if it transforms into large cells and progresses to the tumor stage from the plaque or patch stage. Before diagnosing ALCL or PTCL, a thorough patient clinical history and physical examination is needed. In the present study, among primary cutaneous lymphomas, the incidence of MF was 30.4%, which was lower than reported for Western and Japanese studies.12,13 The reason for the relatively low incidence of MF in our study was probably due a result of not considering patient's clinical findings. Specifically, some PTCL cases may have belonged to the tumor stage of MF. Of the CD30+ LPD cases, there was disagreement with respect to LyP and ALCL between pathologists. Indeed, distinction of LyP from ALCL requires careful evaluation of clinical parameters and relative percentages of large atypical cells, which is a subjective parameter.
- We found that there was a significant difference in incidence compared with Western countries, especially concerning NK/T cell lymphoma and SPTCL. These differences in incidences between countries were very likely the result of viral infection, especially Epstein-Barr virus (EBV) and human T-lymphotropic virus (HTLV). Indeed, EBV positivity is the most important finding in diagnosing NK/T cell lymphoma. Likewise, adult T cell leukemia/lymphoma is associated with HTLV infection and is endemic in several areas of the world, especially in Japan,14 but was rarely observed in the present study.
- Two possibilities for the rarity of the incidence of the PTCL subtype can be suggested. First, this study selected cases from 2006. Compared with the 2001 WHO classification, primary cutaneous aggressive epidermotropic CD8-positive cytotoxic T-cell lymphoma, primary cutaneous γδ T-cell lymphoma, and primary cutaneous small/medium CD4-positive T-cell lymphoma were included as a provisional entity in the new WHO-EORTC classification. Thus, the hemato/dermatopathologist would likely miss these cases. Second, it was difficult to differentiate the PTCL subtype from other cutaneous T-cell lymphomas. For example, primary cutaneous gamma-delta T-cell lymphoma was previously described as SPTCL with a gamma/delta phenotype that shares common morphologic and immunophenotype features. The most important immunophenotype marker for this type of disease is TCRδ, for which detection has recently become available. As such, negativity for βF1 favors a diagnosis of gamma/delta T-cell lymphoma if TCRδ is not appropriate.15 More immunohistochemical staining results for βF1 and TCRδ are needed in cases of PTCL and SPTCL.
- Subtypes of DLBCL were rarely observed in this study. Primary cutaneous DLBCL, leg type, presents on the leg, although other cutaneous sites may be involved. Furthermore, tumor cells of DLBCL, leg type, typically express Bcl2, Bcl6, and MUM1, although approximately 10% of cases do not express either Bcl2 or MUM1.16 By definition, DLBCL comprises all other cases other than those belonging to DLBCL, leg type, and to date only one marker, Bcl6, has been found to be useful in distinguishing between these two subtypes. One study investigated chromosomal aberrations in two subtypes of DLBCL and found similar aberrations irrespective of anatomical site, cell morphology, and Bcl2 expression.17 Among cases of cutaneous B-cell lymphoma, marginal zone B-cell lymphoma was relatively common while follicular lymphoma was rare; these findings were correlated with the results of previous lymphoma studies performed on Korean1 and Japanese populations.13
- In addition PTCL and DLBCL subtypes, it is also important to pay attention to BPDC neoplasm, which is newly described and included in 4th edition of WHO classification. BPDC neoplasm is a subset of acute myeloid leukemia, related to precursor neoplasms,4 and is a clinically aggressive tumor derived from the precursors of plasmacytoid dendritic cells and characterized by a diffuse infiltration of blastic cells with irregular nuclei, fine chromatin and small nucleoli. BPDC neoplasm frequently involves the skin, and has also been described as blastic NK-cell lymphoma.2 Immunohistochemical staining for CD4, CD56, and CD123 is essential to confirm the diagnosis.
- Regretfully, we were unable to evaluate patient outcome associated with the new WHO-EORTC classification system. Thus, further studies concerning patient prognosis and efficacy of different treatment modalities are needed. However, our data were collected from many hospitals where hematopathologists involved in a large number of cases performed the reviews, and included all kinds of cutaneous lymphoma subtypes. In spite of limitations such as absence of comprehensive review of submitted cases, this study may represent the current status of cutaneous lymphoma subtypes in Korea according to new WHO-EORTC classification.
- In conclusion, compared with Western countries, mature T-cell lymphoma was found to be more frequent in Korean patients because of the frequent occurrence of CD30+ LPD, SPTCL, and NK/T cell lymphoma. Conversely, there were fewer cases of B-cell lymphoma, especially follicular lymphomas. The new WHO-EORTC classification system is expected to contribute to uniform diagnosis in cutaneous lymphoma, and thus pathologists and dermatologists should recognize and adapt their diagnoses accordingly.
DISCUSSION
Acknowledgments
Acknowledgments
- 1. Kim JM, Ko YH, Lee SS, et al. WHO classification of malignant lymphomas in Korea: report of the third nationwide study. Korean J Pathol 2011; 45: 254-260. Article
- 2. Leboit PE, Burg G, Weedon D, Sarasin A. Pathology and genetics of skin tumours. Lyon: IARC Press, 2006.
- 3. Willemze R, Jaffe ES, Burg G, et al. WHO-EORTC classification for cutaneous lymphomas. Blood 2005; 105: 3768-3785. ArticlePubMed
- 4. Swerdlow SH, Campo E, Harris NL, et al. WHO classification of tumours of haematopoietic and lymphoid tissues. 4th ed. Lyon: IARC Press, 2008.
- 5. Bradford PT, Devesa SS, Anderson WF, Toro JR. Cutaneous lymphoma incidence patterns in the United States: a population-based study of 3884 cases. Blood 2009; 113: 5064-5073. ArticlePubMedPMCPDF
- 6. Dores GM, Anderson WF, Devesa SS. Cutaneous lymphomas reported to the National Cancer Institute's surveillance, epidemiology, and end results program: applying the new WHO-European Organisation for Research and Treatment of Cancer classification system. J Clin Oncol 2005; 23: 7246-7248. PubMed
- 7. Lee MW. Korean Dermatopathology Research Group. Characteristics of cutaneous lymphomas in Korea. Clin Exp Dermatol 2003; 28: 639-646. ArticlePubMedPDF
- 8. Criscione VD, Weinstock MA. Incidence of cutaneous T-cell lymphoma in the United States, 1973-2002. Arch Dermatol 2007; 143: 854-859. ArticlePubMed
- 9. Morton LM, Turner JJ, Cerhan JR, et al. Proposed classification of lymphoid neoplasms for epidemiologic research from the Pathology Working Group of the International Lymphoma Epidemiology Consortium (InterLymph). Blood 2007; 110: 695-708. PubMedPMC
- 10. Boren E, Gershwin ME. Inflamm-aging: autoimmunity, and the immune-risk phenotype. Autoimmun Rev 2004; 3: 401-406. PubMed
- 11. Riou-Gotta MO, Fournier E, Mermet I, et al. Primary cutaneous lymphomas: a population-based descriptive study of 71 consecutive cases diagnosed between 1980 and 2003. Leuk Lymphoma 2008; 49: 1537-1544. PubMed
- 12. Fink-Puches R, Zenahlik P, Bäck B, Smolle J, Kerl H, Cerroni L. Primary cutaneous lymphomas: applicability of current classification schemes (European Organization for Research and Treatment of Cancer, World Health Organization) based on clinicopathologic features observed in a large group of patients. Blood 2002; 99: 800-805. PubMed
- 13. Fujita A, Hamada T, Iwatsuki K. Retrospective analysis of 133 patients with cutaneous lymphomas from a single Japanese medical center between 1995 and 2008. J Dermatol 2011; 38: 524-530. PubMed
- 14. Lymphoma Study Group of Japanese Pathologists. The World Health Organization classification of malignant lymphomas in Japan: incidence of recently recognized entities. Pathol Int 2000; 50: 696-702. PubMedPDF
- 15. Willemze R, Jansen PM, Cerroni L, et al. Subcutaneous panniculitis-like T-cell lymphoma: definition, classification, and prognostic factors: an EORTC Cutaneous Lymphoma Group Study of 83 cases. Blood 2008; 111: 838-845. PubMed
- 16. Kodama K, Massone C, Chott A, Metze D, Kerl H, Cerroni L. Primary cutaneous large B-cell lymphomas: clinicopathologic features, classification, and prognostic factors in a large series of patients. Blood 2005; 106: 2491-2497. PubMed
- 17. Wiesner T, Streubel B, Huber D, Kerl H, Chott A, Cerroni L. Genetic aberrations in primary cutaneous large B-cell lymphoma: a fluorescence in situ hybridization study of 25 cases. Am J Surg Pathol 2005; 29: 666-673. PubMed
REFERENCES
M, male; F, female; MF, mycosis fungoides; CD30, CD30+ T-cell lymphoproliferative disorder; SPTCL, subcutaneous panniculitis-like T cell lymphoma; PTL, peripheral T cell lymphoma; NK/T, NK/T cell lymphoma, nasal type; AITL, angioimmunoblastic T cell lymphoma; MZBL, marginal zone B-cell lymphoma; DLBCL, diffuse large B-cell lymphoma; BPDCN, blastic plasmacytoid dendritic cell neoplasm; LBL, lymphoblastic lymphoma.
MF, mycosis fungoides; CD30, CD30+ T-cell lymphoproliferative disorder; SPTCL, subcutaneous panniculitis-like T cell lymphoma; PTL, peripheral T cell lymphoma; NK/T, NK/T cell lymphoma, nasal type; AITL, angioimmunoblastic T cell lymphoma; MZBL, marginal zone B-cell lymphoma; DLBCL, diffuse large B-cell lymphoma; BPDCN, blastic plasmacytoid dendritic cell neoplasm; LBL, lymphoblastic lymphoma.
MF, mycosis fungoides; SS, Sezary syndrome; CD30, CD30+ T-cell lymphoproliferative disorder; SPTCL, subcutaneous panniculitis-like T cell lymphoma; PTL, peripheral T cell lymphoma; NK/T, NK/T cell lymphoma, nasal type; AITL, angioimmunoblastic T cell lymphoma; MZBL, marginal zone B-cell lymphoma; FL, follicular lymphoma; DLBCL, diffuse large B-cell lymphoma; BPDCN, blastic plasmacytoid dendritic cell neoplasm; LBL, lymphoblastic lymphoma.
Figure & Data
References
Citations

- Varied presentations of primary cutaneous lymphoma: A case series from a tertiary care center in South India
Baby Shana, Betsy Ambooken, Sunitha Balakrishnan, Asokan Neelakandan, Kidangazhiyathmana Ajithkumar
Indian Journal of Cancer.2024; 61(1): 172. CrossRef - A retrospective study of prognostic factors and treatment outcome in advanced-stage Mycosis Fungoides and Sezary Syndrome
Zhuo-fan Xu, Hongyun Chen, Yuehua Liu, Wei Zhang, Hongzhong Jin, Jie Liu
Hematology.2024;[Epub] CrossRef - Prevalence, clinical features, and survival outcome trends of 627 patients with primary cutaneous lymphoma over 29 years: a retrospective review from single tertiary center in Korea
Ik Jun Moon, Chong Hyun Won, Sung Eun Chang, Chan-Sik Park, Dok-Hyun Yoon, Si Yeol Song, Mi Woo Lee, Woo Jin Lee
Scientific Reports.2024;[Epub] CrossRef - The First Case of Acute Myeloid Leukemia With t(10;11)(p13;q21);PICALM-MLLT10 Rearrangement Presenting With Extensive Skin Involvement
Min-Seung Park, Hyun-Young Kim, Jae Joon Lee, Duck Cho, Chul Won Jung, Hee-Jin Kim, Sun-Hee Kim
Annals of Laboratory Medicine.2023; 43(3): 310. CrossRef - Recent advances on cutaneous lymphoma epidemiology
G. Dobos, M. Miladi, L. Michel, C. Ram-Wolff, M. Battistella, M. Bagot, A. de Masson
La Presse Médicale.2022; 51(1): 104108. CrossRef - Specific cutaneous infiltrates in patients with haematological neoplasms: a retrospective study with 49 patients
Rebeca Calado, Maria Relvas, Francisca Morgado, José Carlos Cardoso, Oscar Tellechea
Australasian Journal of Dermatology.2021;[Epub] CrossRef - Epidemiology of Cutaneous T-Cell Lymphomas: A Systematic Review and Meta-Analysis of 16,953 Patients
Gabor Dobos, Anne Pohrt, Caroline Ram-Wolff, Céleste Lebbé, Jean-David Bouaziz, Maxime Battistella, Martine Bagot, Adèle de Masson
Cancers.2020; 12(10): 2921. CrossRef - Primary cutaneous lymphoma in Argentina: a report of a nationwide study of 416 patients
Alejandra Abeldaño, Paula Enz, Matias Maskin, Andrea B. Cervini, Natallia Torres, Ana C. Acosta, Marina Narbaitz, Silvia Vanzulli, Mirta Orentrajch, Marta A. Villareal, Maria L. Garcia Pazos, Mariana Arias, Evelyn A. Zambrano Franco, Maria I. Fontana, Rob
International Journal of Dermatology.2019; 58(4): 449. CrossRef - Post-thymic CD4 positive cytotoxic T cell infiltrates of the skin: A clinical and histomorphologic spectrum of the unique CD4 positive T cell of immunosenescence
Cynthia M. Magro, Luke C. Olson, Shabnam Momtahen
Annals of Diagnostic Pathology.2019; 38: 99. CrossRef - Cutaneous lymphomas in Taiwan: A review of 118 cases from a medical center in southern Taiwan
Chaw-Ning Lee, Chao-Kai Hsu, Kung-Chao Chang, Cheng-Lin Wu, Tsai-Yun Chen, Julia Yu-Yun Lee
Dermatologica Sinica.2018; 36(1): 16. CrossRef - Imaging analysis of superficial soft tissue lymphomas
In Sook Lee, You Seon Song, Seung Hyun Lee, Young Jin Choi, Sung Moon Lee
Clinical Imaging.2018; 49: 111. CrossRef - Epidemiologic, clinical and demographic features of primary cutaneous lymphomas in Castilla‐La Mancha, Spain: are we different?
C. Ramos‐Rodríguez, M. García‐Rojo, G. Romero‐Aguilera, M. García‐Arpa, L. González‐López, M.P. Sánchez‐Caminero, J. González‐García, M. Delgado‐Portela, M.P. Cortina‐De La Calle, M.F. Relea‐Calatayud, F. Martín‐Dávila, R. López‐Pérez, M. Ramos‐Rodríguez
Journal of the European Academy of Dermatology and Venereology.2018;[Epub] CrossRef - Nasal-type NK/T-cell lymphomas are more frequently T rather than NK lineage based on T-cell receptor gene, RNA, and protein studies: lineage does not predict clinical behavior
Mineui Hong, Taehee Lee, So Young Kang, Suk-Jin Kim, Wonseog Kim, Young-Hyeh Ko
Modern Pathology.2016; 29(5): 430. CrossRef - Cutaneous lymphoma: Kids are not just little people
Katalin Ferenczi, Hanspaul S. Makkar
Clinics in Dermatology.2016; 34(6): 749. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
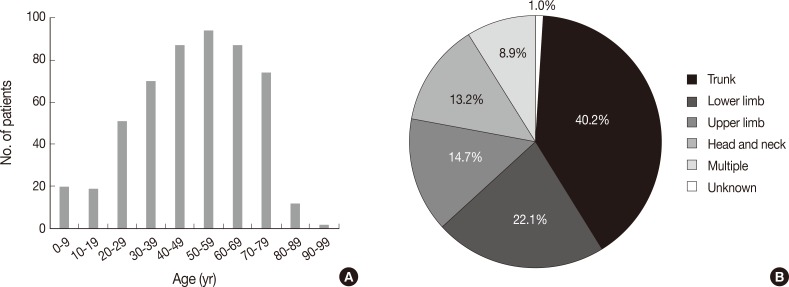
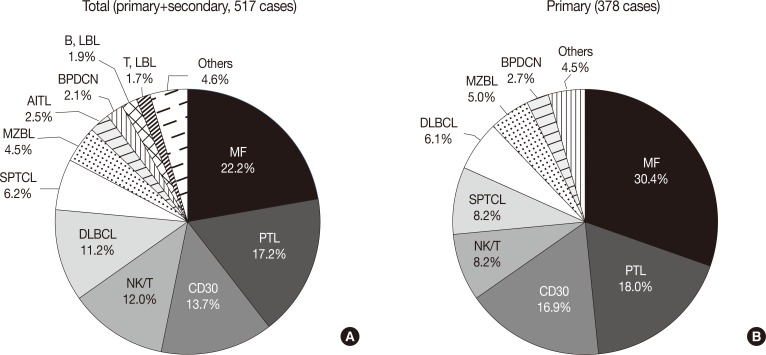
Fig. 1
Fig. 2
| Total | Primary | Secondary | |
|---|---|---|---|
| Mature T-cell lymphoma | |||
| Mycosis fungoides | 110 (21.3) | 110 (29.1) | - |
| Pagetoid reticulosis | 1 (0.2) | 1 (0.3) | - |
| Follicular, syringotropic, granulomatous variants | 3 (0.6) | 3 (0.8) | - |
| Granulomatous slack skin | 1 (0.2) | 1 (0.3) | - |
| Sezary syndrome | 1 (0.2) | 1 (0.3) | - |
| CD30+ T-cell lymphoproliferative disorders | 3 (0.6) | 1 (0.3) | 2 (1.5) |
| Lymphomatoid papulosis | 32 (6.2) | 32 (8.5) | - |
| Anaplastic large cell lymphoma | 36 (7.0) | 31 (8.2) | 5 (3.7) |
| Subcutaneous panniculitis-like T-cell lymphoma | 32 (6.2) | 31 (8.2) | 1 (0.8) |
| Peripheral T-cell lymphoma, unspecified | 71 (13.7) | 51 (13.5) | 19 (14.2) |
| Aggressive epidermotropic CD8-positive cytotoxic T-cell lymphoma | 4 (0.8) | 4 (1.1) | - |
| Gamma/delta-positive T-cell lymphoma | 9 (1.7) | 8 (2.1) | 1 (0.8) |
| Small/medium CD4+ T-cell lymphoma | 5 (1.0) | 5 (1.3) | - |
| Extranodal NK/T cell lymphoma | 58 (11.2) | 27 (7.1) | 30 (22.4) |
| Hydroa vacciniforme-like lymphoma | 4 (0.8) | 4 (1.1) | - |
| Adult T-cell leukemia/lymphoma | 1 (0.2) | - | - |
| Angioimmunoblastic T-cell lymphoma | 13 (2.5) | 1 (0.3) | 12 (9.0) |
| Mature B-cell lymphoma | |||
| Marginal zone B-cell lymphoma | 23 (4.5) | 19 (5.0) | 4 (3.0) |
| Follicle centre lymphoma | 4 (0.8) | 4 (1.1) | - |
| Diffuse large B-cell lymphoma | 43 (8.3) | 17 (4.5) | 26 (19.4) |
| Diffuse large B-cell lylmphoma, leg type | 9 (1.7) | 6 (1.6) | 3 (2.2) |
| Diffuse large B-cell lymphoma, other | 5 (1.0) | - | 4 (3.0) |
| Plasmablastic lymphoma | 1 (0.2) | - | 1 (0.8) |
| Intravascular large B-cell lymphoma | 4 (0.8) | 2 (0.5) | 2 (1.5) |
| Lymphomatoid granulomatosis | 1 (0.2) | 1 (0.3) | - |
| Chronic lymphocytic leukemia | 1 (0.2) | - | 1 (0.8) |
| Burkitt lymphoma | 2 (0.4) | - | 2 (1.5) |
| Immature hematopoietic malignancies | |||
| Blastic plasmacytoid dendritic cell neoplasm | 11 (2.1) | 10 (2.7) | 1 (0.8) |
| T-lymphoblastic lymphoma/leukemia | 9 (1.7) | 2 (0.5) | 7 (5.2) |
| B-lymphoblastic lymphoma/leukemia | 10 (1.9) | 3 (0.8) | 7 (5.2) |
| Hodgkin’s lymphoma | 1 (0.2) | - | 1 (0.8) |
| Others | 9 (1.7) | 3 (0.8) | 6 (4.5) |
| No. of patients (%) | Sex (M/F) | Age distribution (rate, %) |
||||
|---|---|---|---|---|---|---|
| 0-20 yr | 21-40 yr | 41-60 yr | >60 yr | |||
| MF | 115 (22.2) | 1.1 | 3.5 | 36.5 | 41.7 | 18.3 |
| CD30 | 71 (13.7) | 1.3 | 21.1 | 21.1 | 21.1 | 21.1 |
| SPTCL | 32 (6.2) | 0.5 | 15.6 | 50.0 | 25.0 | 9.4 |
| PTL | 89 (17.2) | 1.1 | 5.6 | 21.3 | 38.2 | 34.8 |
| NK/T | 62 (12.0) | 1.5 | 11.3 | 16.1 | 29.0 | 43.5 |
| AITL | 13 (2.5) | 5.5 | 0.0 | 0.0 | 23.1 | 76.9 |
| MZBL | 23 (4.5) | 1.9 | 0.0 | 21.7 | 60.9 | 17.4 |
| DLBCL | 58 (11.2) | 1.9 | 0.0 | 8.6 | 29.3 | 62.1 |
| BPDCN | 11 (2.1) | 10.0 | 9.1 | 36.4 | 9.1 | 45.5 |
| T, LBL | 9 (1.7) | 8.0 | 11.1 | 44.4 | 44.4 | 0.0 |
| B, LBL | 10 (1.9) | 0.7 | 80.0 | 0.0 | 10.0 | 10.0 |
| Others | 24 (4.7) | 2.0 | 4.2 | 0.0 | 33.3 | 62.5 |
| No. of patients (%) | Anatomic location (rate, %) |
|||||
|---|---|---|---|---|---|---|
| Head and neck | Trunk | Upper limb | Lower limb | Multiple | ||
| MF | 115 (22.2) | 4.4 | 53.5 | 7.0 | 16.7 | 18.4 |
| CD30 | 71 (13.7) | 8.6 | 28.6 | 28.6 | 27.1 | 7.1 |
| SPTCL | 32 (6.2) | 3.1 | 53.1 | 12.5 | 12.5 | 18.8 |
| PTL | 89 (17.2) | 15.9 | 37.5 | 13.6 | 26.1 | 6.8 |
| NK/T | 62 (12.0) | 6.5 | 30.6 | 17.7 | 38.7 | 6.5 |
| AITL | 13 (2.5) | 30.8 | 46.2 | 23.1 | 0.0 | 0.0 |
| MZBL | 23 (4.5) | 52.2 | 39.1 | 8.7 | 0.0 | 0.0 |
| DLBCL | 58 (11.2) | 12.5 | 42.9 | 21.4 | 21.4 | 1.8 |
| BPDCN | 11 (2.1) | 27.3 | 36.4 | 9.1 | 18.2 | 9.1 |
| T, LBL | 9 (1.7) | 33.3 | 33.3 | 0.0 | 22.2 | 11.1 |
| B, LBL | 10 (1.9) | 60.0 | 10.0 | 10.0 | 20.0 | 0.0 |
| Others | 24 (4.7) | 12.5 | 45.8 | 8.3 | 29.2 | 4.2 |
| Western (%)5 | Present (%) | |
|---|---|---|
| Mature T-cell lymphoma | 71.3 | 82.3 |
| MF | 38.3 | 30.4 |
| SS | 0.8 | 0.3 |
| CD30 | 10.2 | 16.9 |
| SPTCL | 0.6 | 8.2 |
| PTL | 20.8 | 18.0 |
| NK/T | 0.3 | 8.2 |
| AITL | 0.2 | 0.3 |
| Mature B-cell lymphoma | 28.5 | 13.0 |
| MZBL | 7.1 | 5.0 |
| FL | 8.5 | 1.1 |
| DLBCL | 11.4 | 6.1 |
| Immature hematologic malignancies | 0.3 | 4.0 |
| BPDCN | 0.2 | 2.7 |
| T, LBL | 0.0 | 0.5 |
| B, LBL | 0.1 | 0.8 |
| Others | 1.5 | 0.8 |
Values are presented as number (%). WHO, World Health Organization; EORTC, European Organization for the Research and Treatment of Cancer; NK, natural killer.
M, male; F, female; MF, mycosis fungoides; CD30, CD30+ T-cell lymphoproliferative disorder; SPTCL, subcutaneous panniculitis-like T cell lymphoma; PTL, peripheral T cell lymphoma; NK/T, NK/T cell lymphoma, nasal type; AITL, angioimmunoblastic T cell lymphoma; MZBL, marginal zone B-cell lymphoma; DLBCL, diffuse large B-cell lymphoma; BPDCN, blastic plasmacytoid dendritic cell neoplasm; LBL, lymphoblastic lymphoma.
MF, mycosis fungoides; CD30, CD30+ T-cell lymphoproliferative disorder; SPTCL, subcutaneous panniculitis-like T cell lymphoma; PTL, peripheral T cell lymphoma; NK/T, NK/T cell lymphoma, nasal type; AITL, angioimmunoblastic T cell lymphoma; MZBL, marginal zone B-cell lymphoma; DLBCL, diffuse large B-cell lymphoma; BPDCN, blastic plasmacytoid dendritic cell neoplasm; LBL, lymphoblastic lymphoma.
MF, mycosis fungoides; SS, Sezary syndrome; CD30, CD30+ T-cell lymphoproliferative disorder; SPTCL, subcutaneous panniculitis-like T cell lymphoma; PTL, peripheral T cell lymphoma; NK/T, NK/T cell lymphoma, nasal type; AITL, angioimmunoblastic T cell lymphoma; MZBL, marginal zone B-cell lymphoma; FL, follicular lymphoma; DLBCL, diffuse large B-cell lymphoma; BPDCN, blastic plasmacytoid dendritic cell neoplasm; LBL, lymphoblastic lymphoma.

 E-submission
E-submission