Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(3); 2013 > Article
-
Case Study
A Case of Multifocal Papillary Thyroid Carcinoma Consisting of One Encapsulated Follicular Variant withBRAF K601E Mutation and Three Conventional Types withBRAF V600E Mutation - Wook Youn Kim, Young Sin Ko, Tae Sook Hwang, Hye Seung Han, So Dug Lim, Wan Seop Kim, Seo Young Oh1
-
Korean Journal of Pathology 2013;47(3):293-298.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.3.293
Published online: June 25, 2013
Department of Pathology, Konkuk University School of Medicine, Seoul, Korea.
1Department of Pathology, Konkuk University Medical Center, Seoul, Korea.
- Corresponding Author: Tae Sook Hwang, M.D. Department of Pathology, Konkuk University School of Medicine, 120-1 Neungdong-ro, Gwangjin-gu, Seoul 143-729, Korea. Tel: +82-2-2030-5641, Fax: +82-2-2030-5629, 'tshwang@kuh.ac.kr'
• Received: June 26, 2012 • Revised: September 27, 2012 • Accepted: October 4, 2012
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Multifocal papillary thyroid carcinoma (mPTC) comprises about 20-30% of PTC. In mPTC, individual tumor foci can be identical or frequently composed of different histological types including follicular, solid, tall-cell or conventional patterns. We report a case of mPTC consisting of one encapsulated follicular variant of papillary thyroid carcinoma (FVPTC) and three conventional PTCs in a 44-year-old woman. This case genetically demonstrates unique features including the simultaneous presence of the BRAF V600E (T1799A) mutation and the BRAF K601E (A1801G) mutation in conventional PTC and FVPTC, respectively.
- A 44-year-old female was referred from a local clinic for a papillary carcinoma of the thyroid that was diagnosed by fine needle aspiration. The patient did not have a past history of neck radiation or a family history of thyroid cancer. Thyroid ultrasonography revealed a spiculated, hypoechoic solid nodule measuring 0.8 cm in diameter with a capsular invasion at the left paraisthmic area of the thyroid. The ultrasonography also revealed three smooth, oval, and slightly hypoechoic solid nodules measuring 2.3 cm, 1.3 cm, and 0.8 cm in diameter at the right upper, middle, and lower lobes, respectively. The lymph nodes at the central and lateral neck were not significantly enlarged. Total thyroidectomy with central node dissection was performed.
- Gross findings
- The specimen consisted of a reddish brown total thyroidectomy tissue. The right thyroid gland measured 4.5×2.3×2.0 cm, and the left thyroid gland measured 4.5×2.3×1.7 cm. Both glands weighed 20 g in total. On the serial cut sections of the right thyroid, there were two ill-defined grayish white solid masses measuring 1.2×0.9×0.6 cm and 2.1×1.9×1.5 cm at the upper and lower poles, respectively. At the right mid pole, there also was a well-defined tan, yellow, solid mass measuring 1.3×1.1×0.8 cm. On the serial cut sections of the left lobe, there was an ill-defined grayish white nodule measuring 0.8×0.6×0.5 cm at the paraisthmic area.
- Microscopic findings
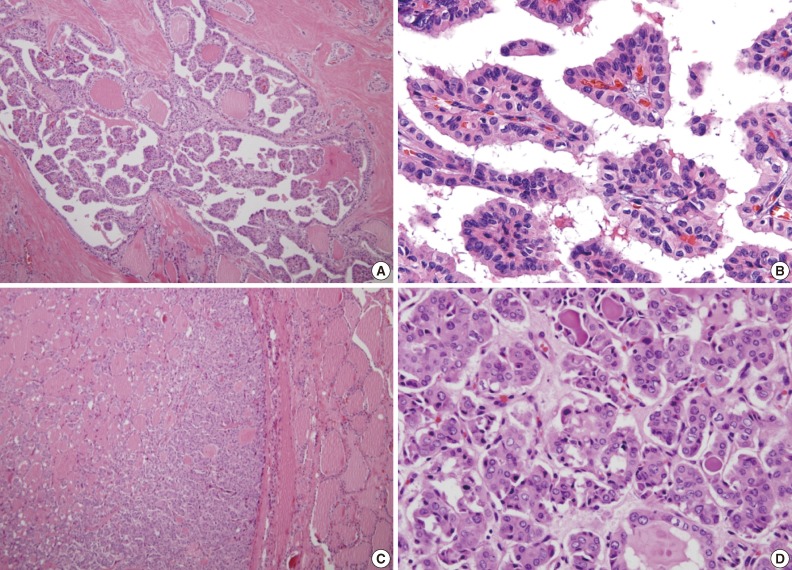
- Histological examination revealed three conventional PTCs in the right upper and lower poles and also in the left paraisthmic area. All the conventional PTCs revealed an infiltrative growth pattern with typical histological features. The mass at the right mid pole was characterized by total encapsulation without papilla formation or psammoma bodies and corresponded to the encapsulated variant of FVPTC. Characteristic nuclear features, particularly nuclear enlargement, chromatin clearing and irregular nuclear contours were present. Nuclear grooves were present, but not frequently. Deeply eosinophilic colloid with peripheral scalloping was also present (Fig. 1). Three perithyroidal lymph nodes were involved by conventional PTC.
- Immunohistochemical findings
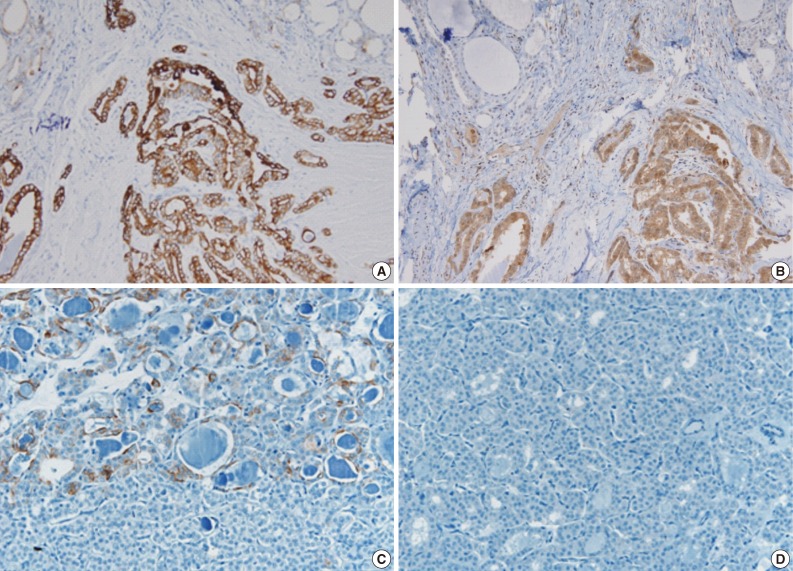
- Immunohistochemical staining was performed on the 5-µm-thick sectioned slides obtained from the formalin-fixed, paraffin embedded tissue using an autoimmunostainer (Ventana, Tucson, AZ, USA) with monoclonal mouse anti-cytokeratin 19 (CK19) antibody (clone A53-B/A2.26, NeoMarkers, Fremont, CA, USA) and polyclonal rabbit anti-galectin-3 antibody (Zymed, San Francisco, CA, USA). The tumor cells of the conventional PTC exhibited a strong, diffusely positive immunoreactivity to CK19 (1:1,500) and galectin 3 (1:100), whereas the tumor cells of the follicular variant revealed no reactivity to galectin 3, and showed only focal and weak staining to CK19 (Fig. 2).
- Molecular analysis
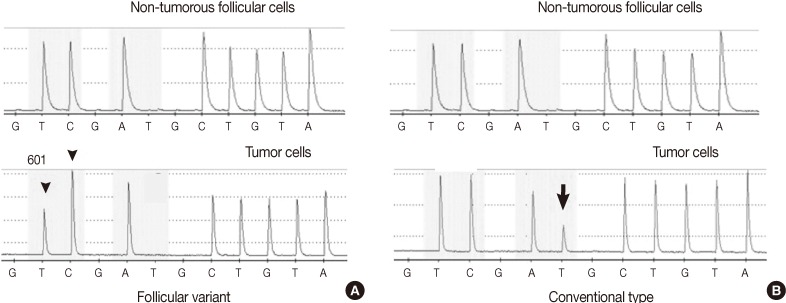
- The tumor cells were micro-dissected from both the conventional and follicular variant areas of the hematoxylin and eosin stained 10 µm-thick sections of the formalin-fixed, paraffin embedded tissue. The genomic DNA was extracted using the extraction buffer solution (50 mM Tris buffer, pH 8.3; 1 mM EDTA, pH 8.0; 5% Tween 20 and 100 µg/mL proteinase K). Mutations of V600E (T1799A) and K601E (A1801G) in exon 15 of the BRAF gene and the codons 12, 13, and 61 of the KRAS and NRAS genes were evaluated by the pyrosequencing method. The DNA extracted from the FVPTC foci revealed a BRAF K601E (A1801G) mutation. The conventional PTCs revealed BRAF V600E (T1799A) mutations in all 3 tumors (Fig. 3). Neither the conventional nor follicular variant revealed KRAS or NRAS mutations.
CASE REPORT
- The most characteristic growth pattern of PTC is papillary, although it is rarely pure and is typically mixed together with a variable proportion of neoplastic follicles. Papillary growth predominates in approximately two thirds of these tumors, whereas another one third is predominantly follicular in characterization. Other histological patterns include solid and trabecular patterns, which are observed in about 20% of cases, but are rarely described as predominate.12 The present case revealed three conventional PTCs and one FVPTC that were distinguished by a complete lack of well-formed papillae, an exclusively microfollicular growth pattern and the characteristic nuclear features of conventional PTC.
- As a mPTC, this case was genetically very unique in that different kinds of BRAF mutations were found simultaneously in accordance with the different histological foci of mPTC. Three conventional PTCs of mPTC had the BRAF V600E (T1799A) mutation and the discrete, noncontiguous FVPTC exhibited the BRAF K601E (A1801G) mutation. To our knowledge, it has never been reported that two different BRAF mutations were noted together in the mPTC of an individual patient at the same time. In previous studies regarding mPTC and its genetic alterations, some cases of mPTC revealed a discordant and mixed pattern of BRAF V600E mutation with a combination of BRAF V600E-positive BRAF V600E-negative PTC foci, although most mPTC exhibited a concordant pattern of either all positive or negative with regards to its BRAF V600E mutation status.5 Since PTC foci that do not harbor the BRAF V600E mutation often show different histopathological types other than the conventional type, BRAF-discordant mPTC tend to show different morphotypes between tumor foci.5 The present case corresponds to the small population of mPTC exhibiting the different morphotype and the discordance of BRAF V600E mutations among discrete tumor foci of mPTC. Moreover, our case was characterized by the presence of the BRAF K601E (A1801G) mutation in BRAF V600E-negative FVPTC foci. If we consider a very low incidence of 7% of BRAF K601E mutations in solitary FVPTC,4 this simultaneous presence of BRAF K601E and V600E mutations must be a very rare event with regards to genetic alterations observed in mPTC. In comparing the frequency of the BRAF K601E mutation with that of the BRAF V600E mutation in FVPTC, the BRAF V600E mutation is more commonly detected in FVPTC due to its high prevalence in PTC. However, the BRAF K601E mutation has special diagnostic value for the detection of FVPTC in that most of this mutation has been encountered and described in FVPTC, although it has been reported in a few cases of follicular adenoma and carcinoma.4,13
- The discordant pattern of BRAF mutations in the present case might be associated with the question whether mPTC arise de novo or from intrathyroidal metastasis. The observed discordant pattern of BRAF mutations in our case seems to support the concept of de novo development of mPTC, suggesting an independent origin of the different tumor foci. However, because there was a previous study that reported that mPTC showing a discordant pattern of BRAF mutations usually has monoclonal derivation when considering the result of genome-wide allelic imbalances,5,11 it remains uncertain whether our case resulted from de novo tumorigenesis or intraglandular spread.
- Immunohistochemical staining showed focal weak positivity to CK19 and no reactivity to galectin 3 in FVPTC, in contrast to diffuse strong reactivity of both makers in conventional PTC. The difference of galectin 3 expression between the conventional type and the follicular variant has been suggested in the previous studies. A large scale comprehensive study reported that the incidence of galectin 3 expression in FVPTC (79.7%) was significantly lower than that of conventional PTC (98.3%),14 although some previous studies on small numbers of FVPTC reported that galectin 3 was expressed in almost all cases like the conventional types.15,16 In terms of CK19 expression, there was no significant difference between FVPTC and the conventional PTC in previous studies.16,17 However, the numbers of FVPTC cases analyzed in these studies on CK19 expression was too small to be generalized. Decreased expression of CK19 and galectin 3 in FVPTC of the present mPTC may reflect the immnohistochemical features of the encapsulated variant of FVPTC.
- Whether the prognosis of mPTC is worse than that of solitary PTC is controversial. Several previous studies have demonstrated that mPTC was associated with a higher rate of recurrence, whereas Schindler et al.18 suggested the opposite.12 The present case consisted of three conventional PTC foci with BRAF V600E mutations and one encapsulated FVPTC foci with the BRAF K601E mutation. Considering that the behavior of encapsulated FVPTC was reported to be indolent in most cases,19 the conventional PTC foci are more likely to affect the clinical course of this patient. In addition, the predominant role of conventional PTC foci in reference to the prognosis might be associated with the fact that BRAF V600E has about 2.5 times the kinase activity of BRAF K601E.20
- In summary, we report a genetically unique case of mPTC having the BRAF V600E (T1799A) mutation in conventional PTC foci and the BRAF K601E (A1801G) mutation in non-contiguous, discrete follicular variant foci, simultaneously.
DISCUSSION
- 1. Liu J, Singh B, Tallini G, et al. Follicular variant of papillary thyroid carcinoma: a clinicopathologic study of a problematic entity. Cancer 2006; 107: 1255–1264. PMID: 16900519. ArticlePubMed
- 2. Xing M. BRAF mutation in thyroid cancer. Endocr Relat Cancer 2005; 12: 245–262. PMID: 15947100. ArticlePubMed
- 3. Kim SK, Hwang TS, Yoo YB, et al. Surgical results of thyroid nodules according to a management guideline based on the BRAF (V600E) mutation status. J Clin Endocrinol Metab 2011; 96: 658–664. PMID: 21239517. ArticlePubMed
- 4. Trovisco V, Vieira de Castro I, Soares P, et al. BRAF mutations are associated with some histological types of papillary thyroid carcinoma. J Pathol 2004; 202: 247–251. PMID: 14743508. ArticlePubMed
- 5. Jovanovic L, Delahunt B, McIver B, et al. Distinct genetic changes characterise multifocality and diverse histological subtypes in papillary thyroid carcinoma. Pathology 2010; 42: 524–533. PMID: 20854070. ArticlePubMed
- 6. Wreesmann VB, Ghossein RA, Hezel M, et al. Follicular variant of papillary thyroid carcinoma: genome-wide appraisal of a controversial entity. Genes Chromosomes Cancer 2004; 40: 355–364. PMID: 15188460. ArticlePubMed
- 7. Sobrinho-Simões M, Preto A, Rocha AS, et al. Molecular pathology of well-differentiated thyroid carcinomas. Virchows Arch 2005; 447: 787–793. PMID: 16189702. ArticlePubMed
- 8. Katoh R, Sasaki J, Kurihara H, Suzuki K, Iida Y, Kawaoi A. Multiple thyroid involvement (intraglandular metastasis) in papillary thyroid carcinoma: a clinicopathologic study of 105 consecutive patients. Cancer 1992; 70: 1585–1590. PMID: 1516009. ArticlePubMed
- 9. Giannini R, Ugolini C, Lupi C, et al. The heterogeneous distribution of BRAF mutation supports the independent clonal origin of distinct tumor foci in multifocal papillary thyroid carcinoma. J Clin Endocrinol Metab 2007; 92: 3511–3516. PMID: 17535994. ArticlePubMed
- 10. Shattuck TM, Westra WH, Ladenson PW, Arnold A. Independent clonal origins of distinct tumor foci in multifocal papillary thyroid carcinoma. N Engl J Med 2005; 352: 2406–2412. PMID: 15944425. ArticlePubMed
- 11. Jovanovic L, Delahunt B, McIver B, Eberhardt NL, Grebe SK. Most multifocal papillary thyroid carcinomas acquire genetic and morphotype diversity through subclonal evolution following the intra-glandular spread of the initial neoplastic clone. J Pathol 2008; 215: 145–154. PMID: 18393366. ArticlePubMed
- 12. Carcangiu ML, Zampi G, Pupi A, Castagnoli A, Rosai J. Papillary carcinoma of the thyroid: a clinicopathologic study of 241 cases treated at the University of Florence, Italy. Cancer 1985; 55: 805–828. PMID: 3967175. ArticlePubMed
- 13. Pennelli G, Vianello F, Barollo S, et al. BRAF(K601E) mutation in a patient with a follicular thyroid carcinoma. Thyroid 2011; 21: 1393–1396. PMID: 22136270. ArticlePubMed
- 14. Jeong JY, Jang JS, Sohn YK, Jung JH, Chun YK, Park JY. Availability of immunohistochemistry in the diagnosis of follicular variant of papillary thyroid carcinoma. Korean J Pathol 2010; 44: 48–55. Article
- 15. Min HS, Choe G, Cho NY, Kang GH, Park SH, Park SY. Immunohistochemical and molecular characteristics of follicular patterned thyroid nodules with incomplete nuclear features of papillary thyroid carcinoma. Korean J Pathol 2009; 43: 495–502. Article
- 16. Zhu X, Sun T, Lu H, et al. Diagnostic significance of CK19, RET, galectin-3 and HBME-1 expression for papillary thyroid carcinoma. J Clin Pathol 2010; 63: 786–789. PMID: 20644217. ArticlePubMed
- 17. Park YJ, Kwak SH, Kim DC, et al. Diagnostic value of galectin-3, HBME-1, cytokeratin 19, high molecular weight cytokeratin, cyclin D1 and p27(kip1) in the differential diagnosis of thyroid nodules. J Korean Med Sci 2007; 22: 621–628. PMID: 17728499. ArticlePubMedPMC
- 18. Schindler AM, van Melle G, Evequoz B, Scazziga B. Prognostic factors in papillary carcinoma of the thyroid. Cancer 1991; 68: 324–330. PMID: 2070332. ArticlePubMed
- 19. Chan J. Strict criteria should be applied in the diagnosis of encapsulated follicular variant of papillary thyroid carcinoma. Am J Clin Pathol 2002; 117: 16–18. PMID: 11791591. ArticlePubMed
- 20. Wan PT, Garnett MJ, Roe SM, et al. Mechanism of activation of the RAF-ERK signaling pathway by oncogenic mutations of B-RAF. Cell 2004; 116: 855–867. PMID: 15035987. ArticlePubMed
References
Fig. 1(A, B) The conventional types in multifocal papillary thyroid carcinoma reveals typical complex and branching papillae with characteristic nuclear features. (C) The follicular variant is encapsulated and composed of small to medium sized, irregularly shaped follicles without papilla formation. (D) Tumor cells of the follicular variant show enlarged, clear nuclei with infrequent nuclear grooves and less irregular nuclear contours compared to conventional foci. Also deeply eosinophilic colloid showing peripheral scalloping is seen within some follicles.
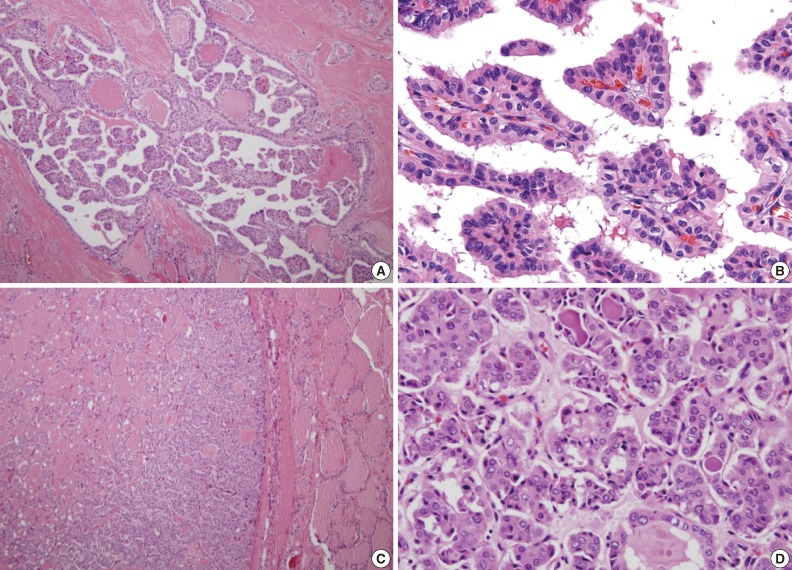

Fig. 2The tumor cells of the conventional papillary thyroid carcinoma (A, B) show strong, diffusely positive immunoreactivity to cytokeratin 19 (A) and galectin 3 (B), whereas the tumor cells of the follicular variant reveal focal, weak positivity to cytokeratin 19 (C), and no reactivity to galectin 3 (D).
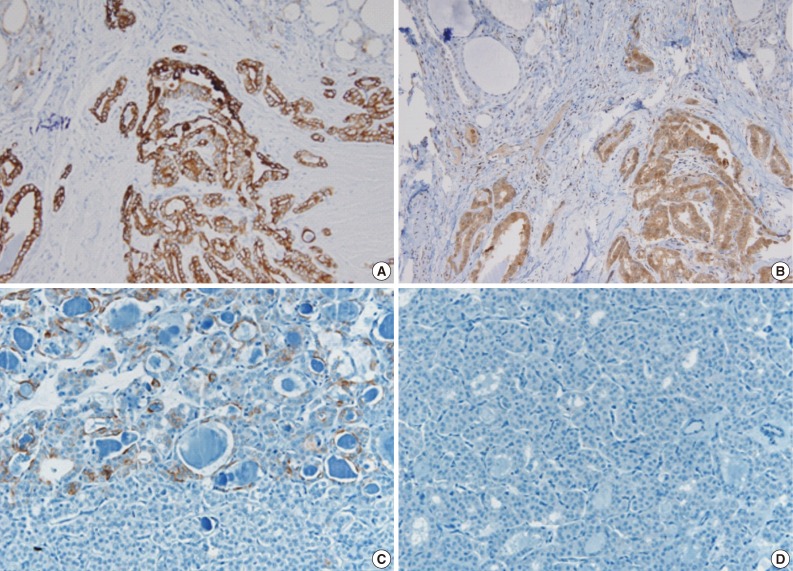

Fig. 3The pyrosequencing analysis of the BRAF mutation shows the BRAF K601E (A1801G) mutation in one follicular variant (A) and the BRAF V600E (T1799A) mutation in three conventional types (B) of multifocal papillary thyroid carcinoma. (A) When the substitution of thymine to cytosine occurs at nucleotide 1801 of exon 15 in the reverse strand, an additional cytosine peak is fused to the next cytosine peak (arrowhead) at nucleotide 1800 with a decrease of the thymine peak at nucleotide 1801 in mutational analysis of the follicular variant. (B) In the three conventional types, the additional thymine peak (arrow) is shown with a decrease of the adenine peak at nucleotide 1799 in reading along the reverse strand.


Figure & Data
References
Citations
Citations to this article as recorded by 

- BRAF K601E Mutation in Oncocytic Carcinoma of the Thyroid: A Case Report and Literature Review
Antonio Matrone, Fabrizia Citro, Carla Gambale, Alessandro Prete, Elisa Minaldi, Raffaele Ciampi, Teresa Ramone, Gabriele Materazzi, Liborio Torregrossa, Rossella Elisei
Journal of Clinical Medicine.2023; 12(22): 6970. CrossRef - Case of aggressive metastatic follicular variant papillary thyroid carcinoma with BRAF K601E and BCORL1 mutations
Doaa Attia, Alexander Lurie, Qihui Zhai, Robert Smallridge
BMJ Case Reports.2020; 13(6): e234208. CrossRef - BRAF gene: From human cancers to developmental syndromes
Muhammad Ramzan Manwar Hussain, Mukhtiar Baig, Hussein Sheik Ali Mohamoud, Zaheer Ulhaq, Daniel C. Hoessli, Ghaidaa Siraj Khogeer, Ranem Radwan Al-Sayed, Jumana Yousuf Al-Aama
Saudi Journal of Biological Sciences.2015; 22(4): 359. CrossRef - Clinical significance of BRAF V600E mutation in 154 patients with thyroid nodules
LINGYING YU, LIZHEN MA, QIAOFENG TU, YI ZHANG, YUEMING CHEN, DAOJUN YU, SHAOYU YANG
Oncology Letters.2015; 9(6): 2633. CrossRef - Clinicopathological Features of Rare BRAF Mutations in Korean Thyroid Cancer Patients
Uiju Cho, Woo Jin Oh, Ja Seong Bae, Sohee Lee, Young Sub Lee, Gyeong Sin Park, Youn Soo Lee, Chan Kwon Jung
Journal of Korean Medical Science.2014; 29(8): 1054. CrossRef - Recurrent Thyroid Papillary Carcinoma in Children Under Ten Years Old: Report of Two Cases and Literature Review
Byeong-Joo Noh, Ji-Youn Sung, Youn-Wha Kim, Yong-Koo Park
Korean Journal of Pathology.2014; 48(4): 297. CrossRef - Anaplastic Transformation of Papillary Thyroid Carcinoma in a Young Man: A Case Study with Immunohistochemical andBRAFAnalysis
Ji Hye Park, Hyeong Ju Kwon, Cheong Soo Park, SoonWon Hong
Korean Journal of Pathology.2014; 48(3): 234. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure

 E-submission
E-submission